The Cole Neurocognition Lab
A cognitive, computational, and network neuroscience laboratory located at the Center for Molecular and Behavioral Neuroscience (CMBN) at Rutgers University
Research Projects
In our research we seek connectivity-based explanations of neurocognitive phenomena, especially those related to learning and goal-directed cognition.
We achieve this using a variety of techniques, applying network science, computational modeling, and machine learning approaches to data collected from the human brain (with fMRI, MEG, EEG, diffusion MRI, and behavioral measures) and neural network simulations.
Much of this work involves understanding the role of brain connectivity in producing the computations apparent in task-driven brain activity patterns and behavior. This facilitates theoretical understanding of cognitive processes as they are generated by brain network interactions, providing insights into both natural and artificial intelligence.
Our ultimate goal is to utilize brain connectivity research to advance fundamental understanding of the human brain, driving applications that enhance the human condition – especially via novel treatments for brain diseases such as major depression, Alzheimer's disease, and schizophrenia.

Neurocognitive Basis of Cognitive Control
Cognitive control consists of the processes underlying goal-directed cognition – processes important for everyday life and disrupted in a variety of mental illnesses. Cognitive control is relevant to most thoughts and behaviors, such as learning, emotion regulation, attention, problem solving, intelligence, and memory.Network Mechanisms of Cognitive Control
-
Ito T, Yang GB, Laurent P, Schultz DH, Cole MW (2022). "Constructing neural network models from brain data reveals representational transformations linked to adaptive behavior". Nature Communications. 13, 673. doi:10.1038/s41467-022-28323-7
Code
On Twitter
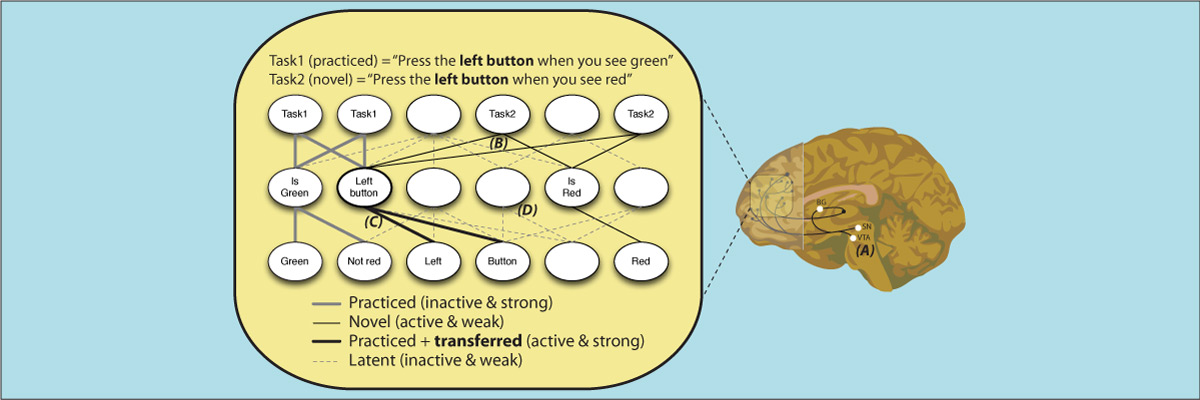
The human ability to adaptively implement a wide variety of tasks is thought to emerge from the dynamic transformation of cognitive information. We hypothesized that these transformations are implemented via conjunctive activations in “conjunction hubs”—brain regions that selectively integrate sensory, cognitive, and motor activations. We used recent advances in using functional connectivity to map the flow of activity between brain regions to construct a task-performing neural network model from fMRI data during a cognitive control task. We verified the importance of conjunction hubs in cognitive computations by simulating neural activity flow over this empirically-estimated functional connectivity model. These empirically-specified simulations produced above-chance task performance (motor responses) by integrating sensory and task rule activations in conjunction hubs. These findings reveal the role of conjunction hubs in supporting flexible cognitive computations, while demonstrating the feasibility of using empirically-estimated neural network models to gain insight into cognitive computations in the human brain.
-
Cocuzza CV, Ito T, Schultz D, Bassett DS, Cole MW (2020). "Flexible coordinator and switcher hubs for adaptive task control". Journal of Neuroscience. 40(36):6949–6968. doi:10.1523/JNEUROSCI.2559-19.2020
On Twitter
On Facebook
Blog Post
Code
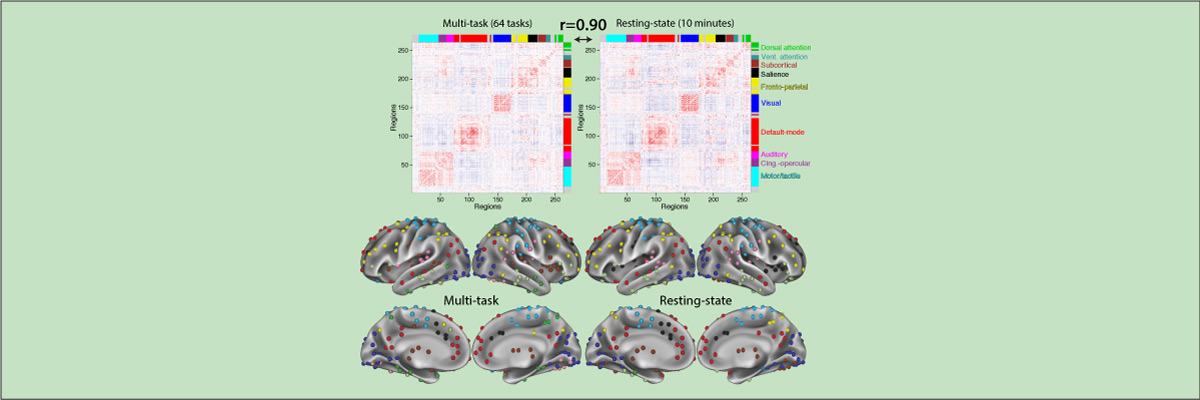
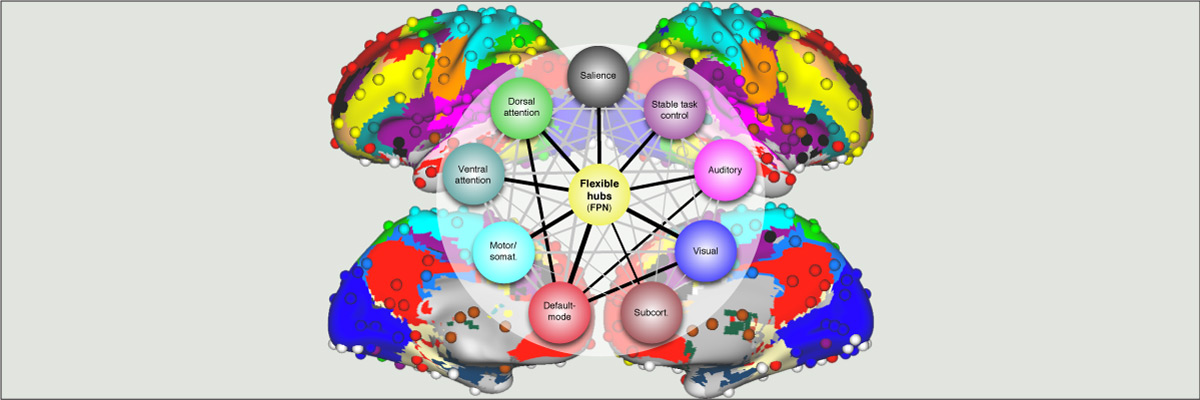
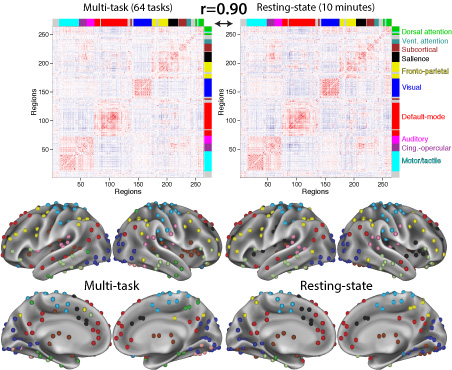
Functional connectivity studies have identified at least two large-scale neural systems that constitute cognitive control networks – the frontoparietal network (FPN) and cingulo-opercular network (CON). Control networks are thought to support goal-directed cognition and behavior. It was previously shown that the FPN flexibly shifts its global connectivity pattern according to task goal, consistent with a “flexible hub” mechanism for cognitive control. Our aim was to build on this finding to develop a functional cartography (a multi-metric profile) of control networks in terms of dynamic network properties. We quantified network properties in (male and female) humans using a high-control-demand cognitive paradigm involving switching among 64 task sets. We hypothesized that cognitive control is enacted by the FPN and CON via distinct but complementary roles reflected in network dynamics. Consistent with a flexible “coordinator” mechanism, FPN connections were globally diverse across tasks, while maintaining within-network connectivity to aid cross-region coordination. Consistent with a flexible “switcher” mechanism, CON regions switched to other networks in a task-dependent manner, driven primarily by reduced within-network connections to other CON regions. This pattern of results suggests FPN acts as a dynamic, global coordinator of goal-relevant information, while CON transiently disbands to lend processing resources to other goal-relevant networks. This cartography of network dynamics reveals a dissociation between two prominent cognitive control networks, suggesting complementary mechanisms underlying goal-directed cognition.
- Cole M.W., Reynolds J.R., Power J.D., Repovs G., Anticevic A., Braver T.S. (2013). "Multi-task connectivity reveals flexible hubs for adaptive task control". Nature Neuroscience; 2013 Sep;16(9):1348-55. doi:10.1038/nn.3470.
More info
Extensive evidence suggests the human ability to adaptively implement a wide variety of tasks is preferentially due to the operation of a fronto-parietal brain network. We hypothesized that this network’s adaptability is made possible by ‘flexible hubs’ – brain regions that rapidly update their pattern of global functional connectivity according to task demands. We utilized recent advances in characterizing brain network organization and dynamics to identify mechanisms consistent with the flexible hub theory. We found that the fronto-parietal network’s brain-wide functional connectivity pattern shifted more than other networks’ across a variety of task states, and that these connectivity patterns could be used to identify the current task. Further, these patterns were consistent across practiced and novel tasks, suggesting reuse of flexible hub connectivity patterns facilitates adaptive (novel) task performance. Together, these findings support a central role for fronto-parietal flexible hubs in cognitive control and adaptive implementation of task demands generally.
- Cole M.W., Yarkoni T., Repovs G., Anticevic A., Braver T.S. (2012). "Global Connectivity of Prefrontal Cortex Predicts Cognitive Control and Intelligence". Journal of Neuroscience. 32(26): 8988-8999; doi: 10.1523/JNEUROSCI.0536-12.2012
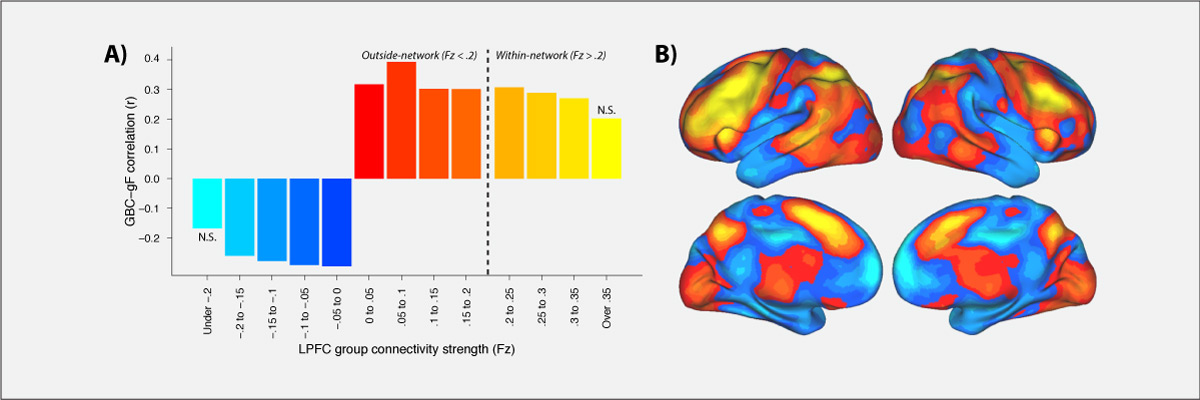
Control of thought and behavior is fundamental to human intelligence. Evidence suggests a frontoparietal brain network implements such cognitive control across diverse contexts. We identify a mechanism — global connectivity — by which components of this network might coordinate control of other networks. A lateral prefrontal cortex (LPFC) region’s activity was found to predict performance in a high control demand working memory task and also to exhibit high global connectivity. Critically, global connectivity in this LPFC region, involving connections both within and outside the frontoparietal network, showed a highly selective relationship with individual differences in fluid intelligence. These findings suggest LPFC is a global hub with a brainwide influence that facilitates the ability to implement control processes central to human intelligence.
- Cole M.W., Schneider W. (2007). “The cognitive control network: Integrated cortical regions with dissociable functions”, NeuroImage 37(1): 343-360. doi: 10.1016/j.neuroimage.2007.03.071
Consensus across hundreds of published studies indicates that the same regions are involved in many forms of cognitive control. Using functional magnetic resonance imaging (fMRI), we found that these coactive regions form a functionally connected cognitive control network (CCN). Network status was identified by convergent methods, including: high interregional correlations during rest and task performance, consistently higher correlations within the CCN than the rest of cortex, co-activation in a visual search task, and mutual sensitivity to decision difficulty. Regions within the CCN include anterior cingulate cortex / pre-supplementary motor area (ACC/pSMA), dorsolateral prefrontal cortex (DLPFC), inferior frontal junction (IFJ), anterior insular cortex (AIC), dorsal pre-motor cortex (dPMC), and posterior parietal cortex (PPC). We used a novel visual line search task which included periods when the probe stimuli were occluded but subjects had to maintain and update working memory in preparation for the sudden appearance of a probe stimulus. The six CCN regions operated as a tightly coupled network during the ‘non-occluded’ portions of this task, with all regions responding to probe events. In contrast, the network was differentiated during occluded search. DLPFC, not ACC/pSMA, was involved in target memory maintenance when probes were absent, while both regions became active in preparation for difficult probes at the end of each occluded period. This approach illustrates one way in which a neuronal network can be identified, its high functional connectivity established, and its components dissociated in order to better understand the interactive and specialized internal mechanisms of that network.
Rapid Instructed Task Learning (RITL; "rittle")
- Cole M.W., Ito T., Braver T.S. (2016). "The behavioral relevance of task information in human prefrontal cortex". Cerebral Cortex. doi: 10.1093/cercor/bhv072
Human lateral prefrontal cortex (LPFC) is thought to play a critical role in enabling cognitive flexibility, particularly when performing novel tasks. However, it remains to be established whether LPFC representation of task-relevant information in such situations actually contributes to successful performance. We utilized pattern classification analyses of functional MRI activity to identify novelty-sensitive brain regions as participants rapidly switched between performance of 64 complex tasks, 60 of which were novel. In three of these novelty-sensitive regions – located within distinct areas of left anterior LPFC – trial-evoked activity patterns discriminated correct from error trials. Further, these regions also contained information regarding the task-relevant decision rule, but only for successfully performed trials. This suggests that left anterior LPFC may be particularly important for representing task information that contributes to the cognitive flexibility needed to perform successfully in novel task situations.
- Cole M.W., Laurent P., Stocco A. (2013). "Rapid instructed task learning: A new window into the human brain’s unique capacity for flexible cognitive control". Cognitive, Affective, & Behavioral Neuroscience. 13(1): 1-22; doi:10.3758/s13415-012-0125-7
The human ability to flexibly adapt to novel circumstances is extraordinary. Perhaps the most illustrative, yet underappreciated, form of this cognitive flexibility is rapid instructed task learning (RITL) – the ability to rapidly reconfigure our minds to perform new tasks from instructions. This ability is important for everyday life (e.g., learning to use new technologies) and is used to instruct participants in nearly every study of human cognition. We review the development of RITL as a circumscribed domain of cognitive neuroscience investigation, culminating in recent demonstrations that RITL is implemented via brain circuits centered on lateral prefrontal cortex. We then build on this and the recent discovery of compositional representations within lateral prefrontal cortex to develop an integrative theory of cognitive flexibility and cognitive control that identifies mechanisms that may enable RITL within the human brain. The insights gained from this new theoretical account have important implications for further developments and applications of RITL research.
- Cole M.W., Bagic A., Kass R., Schneider W. (2010). "Prefrontal Dynamics Underlying Rapid Instructed Task Learning Reverse With Practice". Journal of Neuroscience 30(42):14245-14254. doi: 10.1523/JNEUROSCI.1662-10.2010.
The ability to rapidly reconfigure our minds to perform novel tasks is important for adapting to an ever-changing world, yet little is understood about its basis in the brain. Further, it is unclear how this kind of task preparation changes with practice. Previous research suggests that prefrontal cortex (PFC) is essential when preparing to perform either novel or practiced tasks. Building upon recent evidence that PFC is organized in an anterior-to-posterior hierarchy, we postulated that novel and practiced task preparation would differentiate hierarchically distinct regions within PFC across time. Specifically, we hypothesized and confirmed using functional MRI and magnetoencephalography with humans that novel task preparation is a bottom-up process that involves lower-level rule representations in dorsolateral PFC (DLPFC) prior to a higher-level rule-integrating task representation in anterior PFC (aPFC). In contrast, we identified a complete reversal of this activity pattern during practiced task preparation. Specifically, we found that practiced task preparation is a top-down process that involves a higher-level rule-integrating task representation (recalled from long-term memory) in aPFC prior to lower-level rule representations in DLPFC. These findings reveal two distinct yet highly inter-related mechanisms for task preparation, one involving task set formation from instructions during rapid instructed task learning and the other involving task set retrieval from long-term memory to facilitate familiar task performance. These two mechanisms demonstrate the exceptional flexibility of human PFC as it rapidly reconfigures cognitive brain networks to implement a wide variety of possible tasks.
Computational Network Neuroscience
This is a broad area that utilizes tools from machine learning and network science to understand the nature of brain information processing and, ultimately, behavior. Behavior emerges from the brain's network architecture and the dynamic symphonies that play out on that architecture. We use functional and structural connectivity approaches to characterize properties of large-scale human brain networks, both at rest and during a wide variety of tasks.Select Publications:
-
Sanchez-Romero R, Ito T, Mill RD, Hanson SJ, Cole MW (2023). "Causally informed activity flow models provide mechanistic insight into network-generated cognitive activations". NeuroImage. 278:120300. doi:10.1016/j.neuroimage.2023.120300
On Twitter
Brain activity flow models estimate the movement of task-evoked activity over brain connections to help explain network-generated task functionality. Activity flow models have been shown to accurately generate task-evoked brain activations across a wide variety of brain regions and task conditions. However, these models have had limited explanatory power, given known issues with causal interpretations of the standard functional connectivity measures used to parameterize activity flow models. We show here that functional/effective connectivity (FC) measures grounded in causal principles facilitate mechanistic interpretation of activity flow models. We progress from simple to complex FC measures, with each adding algorithmic details reflecting causal principles. This reflects many neuroscientists’ preference for reduced FC measure complexity (to minimize assumptions, minimize compute time, and fully comprehend and easily communicate methodological details), which potentially trades off with causal validity. We start with Pearson correlation (the current field standard) to remain maximally relevant to the field, estimating causal validity across a range of FC measures using simulations and empirical fMRI data. Finally, we apply causal-FC-based activity flow modeling to a dorsolateral prefrontal cortex region (DLPFC), demonstrating distributed causal network mechanisms contributing to its strong activation during a working memory task. Notably, this fully distributed model is able to account for DLPFC working memory effects traditionally thought to rely primarily on within-region (i.e., not distributed) recurrent processes. Together, these results reveal the promise of parameterizing activity flow models using causal FC methods to identify network mechanisms underlying cognitive computations in the human brain.
-
Mill RD, Hamilton JL, Winfield EC, Lalta N, Chen RH, Cole MW (2022). "Network modeling of dynamic brain interactions predicts emergence of neural information that supports human cognitive behavior". PLOS Biology. doi:10.1101/2021.01.26.428276
Commentary on paper
Code
On Twitter
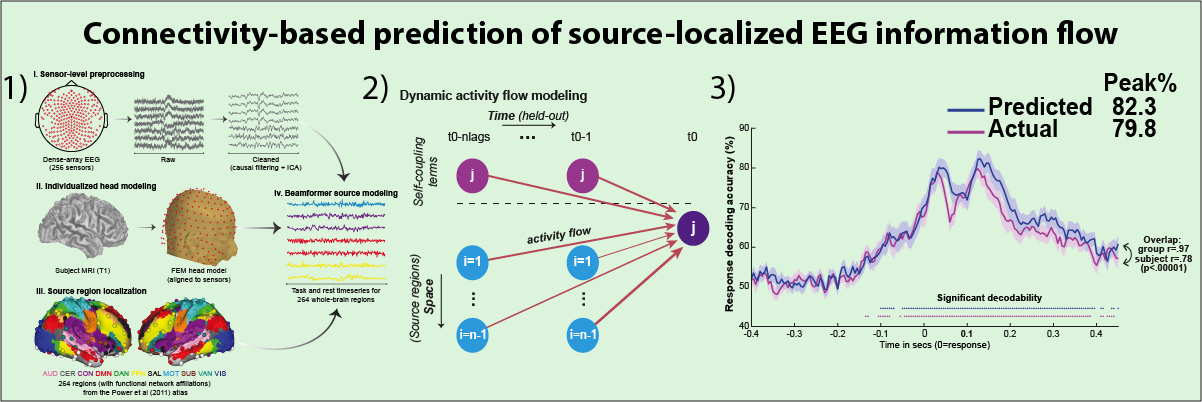
How cognitive task behavior is generated by brain network interactions is a central question in neuroscience. Answering this question calls for the development of novel analysis tools that can firstly capture neural signatures of task information with high spatial and temporal precision (the “where and when”) and then allow for empirical testing of alternative network models of brain function that link information to behavior (the “how”). We outline a novel network modeling approach suited to this purpose that is applied to noninvasive functional neuroimaging data in humans. We first dynamically decoded the spatiotemporal signatures of task information in the human brain by combining MRI-individualized source electroencephalography (EEG) with multivariate pattern analysis (MVPA). A newly developed network modeling approach—dynamic activity flow modeling—then simulated the flow of task-evoked activity over more causally interpretable (relative to standard functional connectivity [FC] approaches) resting-state functional connections (dynamic, lagged, direct, and directional). We demonstrate the utility of this modeling approach by applying it to elucidate network processes underlying sensory–motor information flow in the brain, revealing accurate predictions of empirical response information dynamics underlying behavior. Extending the model toward simulating network lesions suggested a role for the cognitive control networks (CCNs) as primary drivers of response information flow, transitioning from early dorsal attention network-dominated sensory-to-response transformation to later collaborative CCN engagement during response selection. These results demonstrate the utility of the dynamic activity flow modeling approach in identifying the generative network processes underlying neurocognitive phenomena.
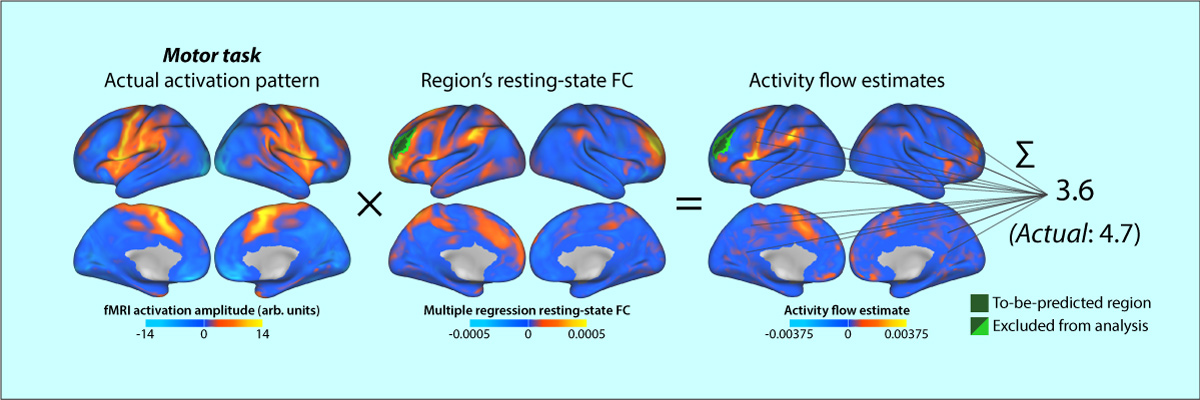
- Cole MW, Ito T, Bassett DS, & Schultz DH (2016). "Activity flow over resting-state networks shapes cognitive task activations". Nature Neuroscience. doi:10.1038/nn.4406
Resting-state functional connectivity (FC) has helped reveal the intrinsic network organization of the human brain, yet its relevance to cognitive task activations has been unclear. Uncertainty remains despite evidence that resting-state FC patterns are highly similar to cognitive task activation patterns. Identifying the distributed processes that shape localized cognitive task activations may help reveal why resting-state FC is so strongly related to cognitive task activations. We found that estimating task-evoked activity flow (the spread of activation amplitudes) over resting-state FC networks allowed prediction of cognitive task activations in a large-scale neural network model. Applying this insight to empirical functional MRI data, we found that cognitive task activations can be predicted in held-out brain regions (and held-out individuals) via estimated activity flow over resting-state FC networks. This suggests that task-evoked activity flow over intrinsic networks is a large-scale mechanism explaining the relevance of resting-state FC to cognitive task activations.
More information on Cole, Ito, et al. (2016):
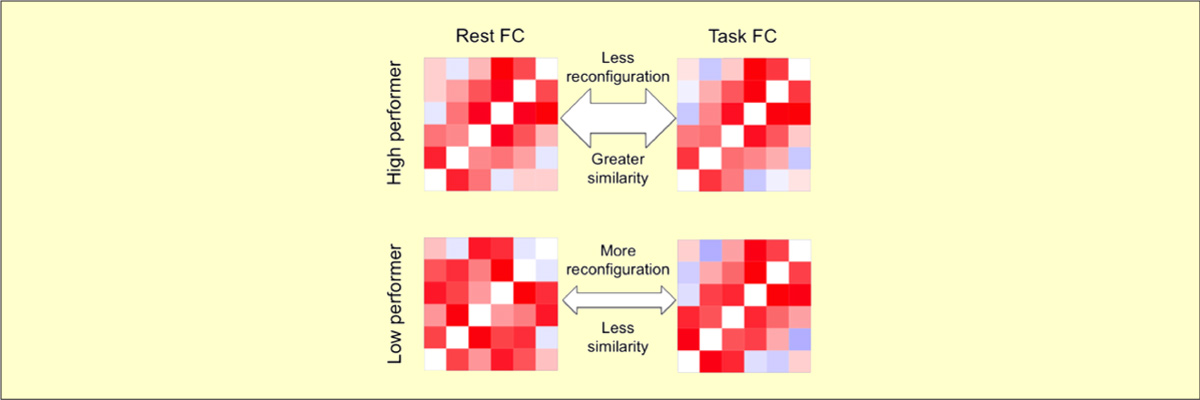
- Schultz DH & Cole MW (2016). "Higher Intelligence Is Associated with Less Task-Related Brain Network Reconfiguration". Journal of Neuroscience. doi:10.1523/JNEUROSCI.0358-16.2016
The human brain is able to exceed modern computers on multiple computational demands (e.g., language, planning) using a small fraction of the energy. The mystery of how the brain can be so efficient is compounded by recent evidence that all brain regions are constantly active as they interact in so-called resting-state networks (RSNs). To investigate the brain's ability to process complex cognitive demands efficiently, we compared functional connectivity (FC) during rest and multiple highly distinct tasks. We found previously that RSNs are present during a wide variety of tasks and that tasks only minimally modify FC patterns throughout the brain. Here, we tested the hypothesis that, although subtle, these task-evoked FC updates from rest nonetheless contribute strongly to behavioral performance. One might expect that larger changes in FC reflect optimization of networks for the task at hand, improving behavioral performance. Alternatively, smaller changes in FC could reflect optimization for efficient (i.e., small) network updates, reducing processing demands to improve behavioral performance. We found across three task domains that high-performing individuals exhibited more efficient brain connectivity updates in the form of smaller changes in functional network architecture between rest and task. These smaller changes suggest that individuals with an optimized intrinsic network configuration for domain-general task performance experience more efficient network updates generally. Confirming this, network update efficiency correlated with general intelligence. The brain's reconfiguration efficiency therefore appears to be a key feature contributing to both its network dynamics and general cognitive ability.
- Cole M.W., Bassett D.S., Power J.D., Braver T.S., Petersen S.E. (2014). "Intrinsic and task-evoked network architectures of the human brain". Neuron. 83(1), 238 – 251. doi:10.1016/j.neuron.2014.05.014
Many functional network properties of the human brain have been identified during rest and task states, yet it remains unclear how the two relate. We identified a whole-brain network architecture present across dozens of task states that was highly similar to the resting-state network architecture. The most frequent functional connectivity strengths across tasks closely matched the strengths observed at rest, suggesting this is an “intrinsic”, standard architecture of functional brain organization. Further, a set of small but consistent changes common across tasks suggests the existence of a task-general network architecture distinguishing task states from rest. These results indicate the brain’s functional network architecture during task performance is shaped primarily by an intrinsic network architecture that is also present during rest, and secondarily by evoked task-general and task-specific network changes. This establishes a strong relationship between resting-state functional connectivity and task-evoked functional connectivity – areas of neuroscientific inquiry typically considered separately.

Network Disruptions Underlying Brain Disorders and Aging
We also investigate how the brain network properties identified in the first two research programs may explain symptoms of brain disorders, as well as effects of (healthy and unhealthy) aging. This works on two levels: First, cognitive control deficits are present across many brain/mental disorders and also emerge with aging. Second, we found that similar brain network disruptions are present across multiple brain disorders. Thus, discoveries in these areas may facilitate understanding of the deficits underlying brain disorders, possibly leading to treatments that apply across multiple disorders (and potentially aging as well).Select Publications:
-
Hearne LJ, Mill RD, Keane BP, Repovs G, Anticevic A, Cole MW (2021). "Activity flow underlying abnormalities in brain activations and cognition in schizophrenia". Science Advances. 7(9) doi:10.1126/sciadv.abf2513
Code
On Twitter
Cognitive dysfunction is a core feature of many brain disorders such as schizophrenia (SZ), and has been linked to both aberrant brain functional connectivity (FC) and aberrant cognitive brain activations. We propose that aberrant network activity flow over FC pathways leads to altered cognitive activations that produce cognitive dysfunction in SZ. We tested this hypothesis using activity flow mapping – an approach that models the movement of task-related activity between brain regions as a function of FC. Using fMRI data from SZ individuals and healthy controls during a working memory task, we found that activity flow models accurately predict aberrant cognitive activations across multiple brain networks. Within the same framework, we simulated a connectivity-based clinical intervention, predicting specific treatments that normalized brain activations and behavior in independent patients. Our results suggest that dysfunctional task-evoked activity flow is a large-scale network mechanism contributing to the emergence of cognitive dysfunction in SZ.
-
Mill RD, Gordon BA, Balota DA, Cole MW (2020). "Predicting dysfunctional age-related task activations from resting-state network alterations". NeuroImage. doi:10.1016/j.neuroimage.2020.117167
On Twitter
Code
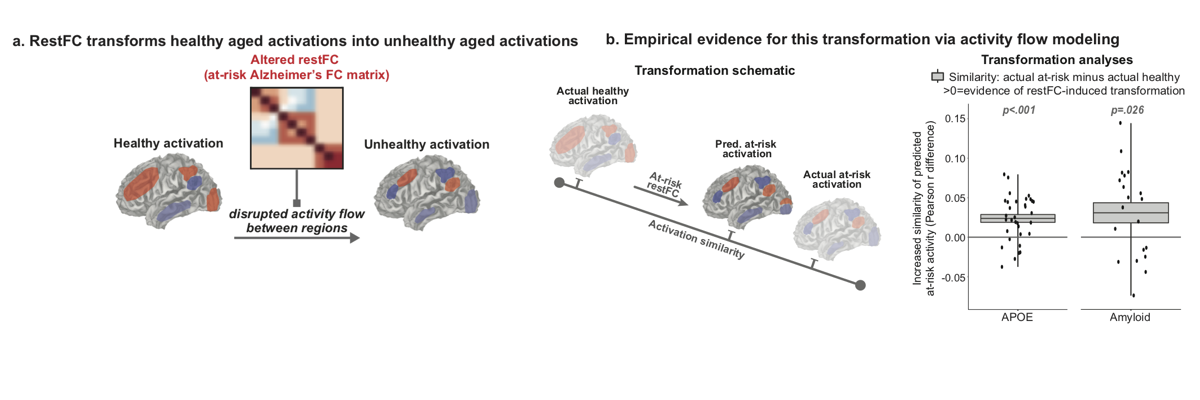
Alzheimer’s disease (AD) is linked to changes in fMRI task activations and fMRI resting-state functional connectivity (restFC), which can emerge early in the timecourse of illness. Study of these fMRI correlates of unhealthy aging has been conducted in largely separate subfields. Taking inspiration from neural network simulations, we propose a unifying mechanism wherein restFC network alterations associated with Alzheimer’s disease disrupt the ability for activations to flow between brain regions, leading to aberrant task activations. We apply this activity flow modeling framework in a large sample of clinically unimpaired older adults, which was segregated into healthy (low-risk) and at-risk subgroups based on established imaging (positron emission tomography amyloid) and genetic (apolipoprotein) risk factors for AD. We identified healthy task activations in individuals at low risk for AD, and then by estimating activity flow using at-risk AD restFC data we were able to predict the altered at-risk AD task activations. Thus, modeling the flow of healthy activations over at-risk AD connectivity effectively transformed the healthy aged activations into unhealthy aged activations. These results provide evidence that activity flow over altered intrinsic functional connections may act as a mechanism underlying Alzheimer’s-related dysfunction, even in very early stages of the illness. Beyond these mechanistic insights linking restFC with cognitive task activations, this approach has potential clinical utility as it enables prediction of task activations and associated cognitive dysfunction in individuals without requiring them to perform in-scanner cognitive tasks.
-
Schultz DH, Ito T, Solomyak L, Chen RH, Mill RD, Anticevic A, Cole MW (2019). "Global connectivity of the frontoparietal cognitive control network is related to depression symptoms in the general population". Network Neuroscience. 3:1:107-123. doi:10.1162/NETN_a_00056
On Twitter
On Facebook
Blog Post
We all vary in our mental health, even among people not meeting diagnostic criteria for mental illness. Understanding this individual variability may reveal factors driving the risk for mental illness, as well as factors driving sub-clinical problems that still adversely affect quality of life. To better understand the large-scale brain network mechanisms underlying this variability we examined the relationship between mental health symptoms and resting-state functional connectivity patterns in cognitive control systems. One such system is the frontoparietal cognitive control network (FPN). Changes in FPN connectivity may impact mental health by disrupting the ability to regulate symptoms in a goal-directed manner. Here we test the hypothesis that FPN dysconnectivity relates to mental health symptoms even among individuals who do not meet formal diagnostic criteria but may exhibit meaningful symptom variation. We found that depression symptoms severity negatively correlated with between-network global connectivity (BGC) of the FPN. This suggests that decreased connectivity between the FPN and the rest of the brain is related to increased depression symptoms in the general population. These findings complement previous clinical studies to support the hypothesis that global FPN connectivity contributes to the regulation of mental health symptoms across both health and disease.
-
Spronk M, Keane BP, Ito T, Kulkarni K, Ji JL, Anticevic A, Cole MW (2021). "A whole-brain and cross-diagnostic perspective on functional brain network dysfunction". Cerebral Cortex. doi:10.1093/cercor/bhaa242
On Twitter
A wide variety of mental disorders have been associated with resting-state functional network alterations, which are thought to contribute to the cognitive changes underlying mental illness. These observations have seemed to support various theories postulating large-scale disruptions of brain systems in mental illness. However, existing approaches isolate differences in network organization without putting those differences in broad, whole-brain perspective. Using a graph distance measure - connectome-wide correlation - we found that whole-brain resting-state functional network organization in humans is highly similar across a variety of mental diseases and healthy controls. This similarity was observed across autism spectrum disorder, attention-deficit hyperactivity disorder, and schizophrenia. Nonetheless, subtle differences in network graph distance were predictive of diagnosis, suggesting that while functional connectomes differ little across health and disease those differences are informative. Such small network alterations may reflect the fact that most psychiatric patients maintain overall cognitive abilities similar to those of healthy individuals (relative to, e.g., the most severe schizophrenia cases), such that whole-brain functional network organization is expected to differ only subtly even for mental diseases with devastating effects on everyday life. These results suggest a need to reevaluate neurocognitive theories of mental illness, with a role for subtle functional brain network changes in the production of an array of mental diseases.
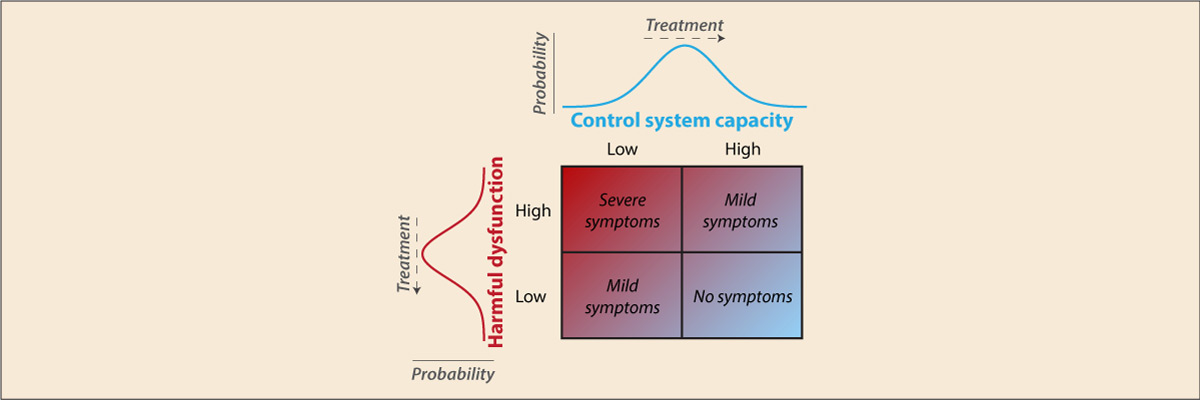
- Cole M.W., Repovs G., Anticevic A. (2014). "The frontoparietal control system: A central role in mental health". The Neuroscientist, 20(6), 652–664. doi:10.1177/1073858414525995
Recent findings suggest the existence of a fronto-parietal control system consisting of flexible hubs that regulate distributed systems (e.g., visual, limbic, motor) according to current task goals. A growing number of studies are reporting alterations of this control system across a striking range of mental diseases. We suggest this may reflect a critical role for the control system in promoting and maintaining mental health. Specifically, we propose that this system implements feedback control to regulate symptoms as they arise (e.g., excessive anxiety reduced via regulation of amygdala), such that an intact control system is protective against a variety of mental illnesses. Consistent with this possibility, recent results indicate that several major mental illnesses involve altered brain-wide connectivity of the control system, likely altering its ability to regulate symptoms. These results suggest that this ‘immune system of the mind’ may be an especially important target for future basic and clinical research.
- Cole M.W., Anticevic A., Repovs G., Barch D. (2011). "Variable global dysconnectivity and individual differences in schizophrenia". Biological Psychiatry 70(1):43-50. doi:10.1016/j.biopsych.2011.02.010
Background
A fundamental challenge for understanding neuropsychiatric disease is identifying sources of individual differences in psychopathology, especially when there is substantial heterogeneity of symptom expression such as is found in schizophrenia. We hypothesized that such heterogeneity may arise in part from consistently widespread yet variably patterned alterations in the connectivity of focal brain regions.
Methods
We used resting state functional MRI to identify variable global dysconnectivity in 23 patients with DSM-IV schizophrenia relative to 22 age, gender, and parental socioeconomic status matched controls using a novel global brain connectivity (GBC) functional MRI method that is robust to high variability across individuals. We examined cognitive functioning using a modified Sternberg task and subtests from the Wechsler Adult Intelligence Scale - Third Edition. We measured symptom severity using the Scale for Assessment of Positive and Negative Symptoms.
Results
We identified a dorsolateral prefrontal cortex (DLPFC) region with global and highly variable dysconnectivity involving within-PFC under-connectivity and non-PFC over-connectivity in patients. Variability in this ‘under/over’ pattern of dysconnectivity strongly predicted the severity of cognitive deficits (matrix reasoning IQ, verbal IQ, and working memory performance) as well as individual differences in every cardinal symptom domain of schizophrenia (poverty, reality distortion, and disorganization).
Conclusion
These results suggest that global dysconnectivity underlies DLPFC involvement in the neuropathology of schizophrenia. Further, these results demonstrate the possibility that specific patterns of dysconnectivity with a given network hub region may explain individual differences in symptom presentation in schizophrenia. Critically, such findings may extend to other neuropathologies with diverse presentation.
News
Job openings
- We welcome undergraduate research volunteers, graduate students, and medical students/fellows interested in cognitive and human brain research. We primarily accept graduate students via the Graduate Program in Neuroscience at Rutgers University-Newark.
We are especially interested in candidates with solid computer science, neuroscience, data science, biology, statistics, or psychology training and excellent programming and quantitative skills. Experience in human brain imaging, computational neuroscience, software engineering, or data science is a plus.
Learn more about the lab
Lab Stats
| Publications | Preprints | Conference Presentations |
|---|
People in the Lab

Michael W. Cole, PhD Principal Investigator
ProfessorCenter for Molecular and Behavioral Neuroscience (CMBN), Rutgers University-Newark
Email: michael.cole@rutgers.edu
Michael's Rutgers University profile
Michael's curriculum vitae
Michael's citations on Google Scholar
Bluesky: @mwcole.bsky.social

Ravi Mill, PhD Research Faculty
PhD: University of St. Andrews in Psychology & Neuroscience (2015)BS: University College London in Psychology (2010)
Email: ravi.mill@rutgers.edu
Twitter: @RaviMill

Ruben Sanchez-Romero, PhD Postdoctoral Fellow
PhD: Carnegie Mellon University (2018)Ruben's citations on Google Scholar
Email: ruben.saro@rutgers.edu

Kirsten Peterson Graduate Student
BS: University of Southern California in Computational Neuroscience (2018)Email: klp173@rutgers.edu

Lakshman Chakravarthula Graduate Student
MS: Indian Institute of Technology Gandhinagar in Cognitive Science (2019)Bachelor of Technology: Indian Institute of Technology Roorkee in Metallurgical and Materials Engineering (2017)

Alexandros Tzalavras Graduate Student
MS: New Jersey Institute of Technology in Biomedical Engineering (2022)BS & MS: National Technical University of Athens (NTUA) – Athens, Greece in Electrical and Computer Engineering (2020)

Arun Aryal Graduate Student
BS: New Jersey Institute of Technology in Biomedical Engineering (2023)
Lily Thompson Graduate Student
BS: Lafayette College in Neuroscience (2024)
Other members of the lab
Gifty Jones Undergraduate Research Assistant
Lab Alumni
Matthew Singh, PhD Postdoctoral Fellow
Luke Hearne, PhD Postdoctoral Fellow
Alisson López-Donado Undergraduate Research Assistant
Shraeyah N. Rajeshwaran Undergraduate Research Assistant
Cindy Hussein Undergraduate Research Assistant
Ella Podvalny Postdoctoral Fellow
Adeola Ajiboro Undergraduate Research Assistant
Temitope F. Ayetan Undergraduate Research Assistant
Carrisa Cocuzza Graduate Student
Micah Ketola Graduate Student
Edwin Lotero Undergraduate Research Assistant
Nicole Lalta Lab Manager & Research Assistant
Aditya Rao Research Assistant
Akshay Warrier Undergraduate Research Assistant
Avi Shah Research Assistant
Brian Keane Visiting Scholar
Jada White Undergraduate Research Assistant
Willio Jeanvilma Undergraduate Research Assistant
Richard Chen Graduate Student
Ethan McCormick Postdoctoral Fellow
Emily Winfield Lab Manager & Research Assistant
Takuya Ito Graduate Student
Diana Rodas Undergraduate Research Assistant
Catalina Guzman Undergraduate Research Assistant
Katelyn Arnemann Postdoctoral Fellow
Julia Hamilton Lab Manager & Research Assistant
Ian Kim Rotation Graduate Student
Marjolein Spronk Postdoctoral fellow
Aisha Assaf Undergraduate Research Assistant
Doug Schultz Postdoctoral fellow
Katherine Wolfert Rotation Graduate Student
Yanira Sanchez Undergraduate Research Assistant
Guochun Yang Visiting Scholar
Kaustubh Kulkarni Lab Manager & Research Assistant
Miguel Vivar Undergraduate Research Assistant
Brayan Zambrono Undergraduate Research Assistant
Levi Solomyak Lab Manager & Research Assistant
Senne Braem Visiting Scholar
Sara Horton Undergraduate Research Assistant
Eryn Fox Research Assistant
Pinelopi Kyriazi Rotation Graduate Student
Mia Tharp Visiting Scholar
Public Information & Outreach
On the Web:
- Practically Scientific - a newsletter about the transformational potential of science & reason, and the cognitive biases holding us back
- Cole Neurocognition Lab's blog – In-depth thoughts on cognitive neuroscience (more extensive that our Facebook and Bluesky posts)
- Neurevolution – A blog dedicated to communicating cognitive neuroscience to a broad audience
In the news:
- "Neuroscientist Michael Cole Uses Machine Learning and Predictive Modeling to Help Patients With Cognitive Deficits" - Rutgers SASN News
- Empirical Neural Networks - Brain Inspired Podcast featuring Michael W. Cole On Twitter
- "New Diagnostic Method May Predict Relapse Risk From Prescription Opioid Addiction" - Rutgers Today
- Interviewed by the Cognitive Neuroscience Society in connection with receiving the 2019 Young Investigator Award:
Going with the Flow: Mapping Information in the Human Brain - Press release from Society for Neuroscience: Higher Intelligence Is Associated with Less Task-Related Brain Network Reconfiguration
- Interviewed for: "Toward Predicting Personalized Neural Responses" – The Scientist
- Interviewed for: "How Relying on Apps to Remember Things Is Making It Easier For Me to Forget Them" – Motherboard
- Interviewed for: "What Happens When an Internet-Connected Brain Has a Stroke?" – Motherboard
- Active or at rest, brain conducts similar symphonies – Simons Foundation Autism Research Initiative
- Brains At Rest Function The Same As Active Brains; Mental Health Research Will Benefit – Medical Daily
- Neuroscience Discovery May Accelerate Brain Research – Psych Central
- Working to Loosen the Grip of Severe Mental Illness – Rutgers Today
- Brain's Flexible Hub Network Helps Humans Adapt – ScienceDaily
- How human brains adapt to new cognitive challenges – DNA India
- A smart hub in the brain – Nature (research highlight)
- Intelligence: Brain size matters, but so do connections – Los Angeles Times
- BBC World Service's Newsday interview – Over 80 million listeners (source)
- Brain imaging can tell how intelligent you are – Times of India
- The Best Way to Measure Intelligence Could Be Brain Imaging – Popular Science
- Imaging global brain connectivity can predict how intelligent you are – Kurzweil News
Publications
Preprints
Chakravarthula LN, Ito T, Tzalavras A, Cole MW (Preprint). "Network geometry shapes multi-task representational transformations across human cortex". bioRxiv. doi:10.1101/2025.03.14.643366
Rosenberg-Lee M, Rennerfeldt P, Abreu-Mendoza RA, Nkwo C, Shaheed P, Coulanges L, Mill R, Cole MW, Pincus M (Preprint). "Whole number interference in rational number processing: Neuroimaging evidence from decimal compariso". PsyArXiv. doi:10.31234/osf.io/uwr7j_v3
Brough R, Chiew KS, Cole MW, Braver T (Preprint). "Deconstructing Proactive Control and Reward Incentive Effects on Rapid Instructed Task Learning". OSF Preprint. doi:10.31219/osf.io/4egtx
Peterson KL, Sanchez-Romero R, Mill RD, Cole MW (Preprint). "Regularized partial correlation provides reliable functional connectivity estimates while correcting for widespread confounding". bioRxiv. doi:10.1101/2023.06.27.546751 On Twitter
Mill RD, Cole MW (Preprint). "Dynamically shifting from compositional to conjunctive brain representations supports cognitive task learning". bioRxiv. doi:10.1101/2023.06.27.546751 On Twitter
2025
Blujus J, Cole MW, Festa EK, Buka SL, Salloway SP, Heindel WC, Oh H (2025). Functional Redundancy of the Posterior Hippocampi is Selectively Disrupted in Non-demented Older Adults with β-amyloid Deposition. NeuroImage: Reports
Singh MF, Braver TS, Cole MW, Ching S (2025). "Precision data-driven modeling of cortical dynamics reveals person-specific mechanisms underpinning brain electrophysiology". PNAS. doi.org/10.1073/pnas.2409577121
Chai M, Palenciano AF, Mill R, Cole MW, Braem S (2025). It's hard to prepare for task novelty: Cueing the novelty of upcoming tasks does not facilitate task performance. Journal of Cognition. doi.org/10.5334/joc.423
2024
Cocuzza CV, Sanchez-Romero R, Ito T, Mill RD, Keane BP, Cole MW (2024). "Distributed network flows generate localized category selectivity in human visual cortex". PLOS Computational Biology. doi:10.1101/2022.02.19.481103 On Bluesky
Cole MW (2024). "Cognitive flexibility as the shifting of brain network flows by flexible neural representations". Current Opinion in Behavioral Sciences. 57:101384. doi.org/10.1016/j.cobeha.2024.101384
Podvalny E, Sanchez-Romero R, Cole MW (2024). "Functionality of arousal-regulating brain circuitry at rest predicts human cognitive abilities". Cerebral Cortex. 34:bhae192. doi.org/10.1093/cercor/bhae192
Cole MW (2024). "Network modeling: The explanatory power of activity flow models of brain function". Chapter in Computational and Network Modeling of Neuroimaging Data.
Keane BP, Abrham Y, Cole MW, Johnson BA, Hu B, Cocuzza CV (2024). "Functional dysconnectivity of visual and somatomotor networks yields a simple and robust biomarker for psychosis". Molecular Psychiatry 10.1038/s41380-024-02767-3
2023
Rosenberg-Lee M, Varma S, Cole MW, Abreu-Mendoza RA (2023). "Competing numerical magnitude codes in decimal comparison: Whole number and rational number distance both impact performance". Cognition. doi:10.1016/j.cognition.2023.105608
Sanchez-Romero R, Ito T, Mill RD, Hanson SJ, Cole MW (2023). "Causally informed activity flow models provide mechanistic insight into network-generated cognitive activations". NeuroImage. 278:120300. doi:10.1016/j.neuroimage.2023.120300 Code On Twitter
Keane BP, Krekelberg B, Mill RD, Silverstein SM, Thompson JL, Serody MR, Barch DM, Cole MW (In Press). "Dorsal attention network activity during perceptual organization is distinct in schizophrenia and predictive of cognitive disorganization". European Journal of Neuroscience. doi:10.1111/ejn.15889 On Twitter
2022
Hwang K, Shine JM, Cole MW, Sorenson E. (2022). "Thalamocortical contributions to cognitive task activity". eLife. doi:10.7554/eLife.81282
Mill RD, Hamilton JL, Winfield EC, Lalta N, Chen RH, Cole MW (2022). "Network modeling of dynamic brain interactions predicts emergence of neural information that supports human cognitive behavior". PLOS Biology. doi:10.1101/2021.01.26.428276 Commentary on paper Code On Twitter
Ito T, Klinger T, Schultz DH, Murray JD, Cole MW, Rigotti M (2022). "Compositional generalization through abstract representations in human and artificial neural networks". NeurIPS Proceedings. doi:10.48550/arXiv.2209.07431
Ito T, Yang GB, Laurent P, Schultz DH, Cole MW (2022). "Constructing neural network models from brain data reveals representational transformations linked to adaptive behavior". Nature Communications. 13, 673. doi:10.1038/s41467-022-28323-7 Code On Twitter
Cocuzza CV, Sanchez-Romero R, Cole MW (2022). "Protocol for activity flow mapping of neurocognitive computations using the Brain Activity Flow Toolbox". STAR Protocols. 3, 1. doi:10.1016/j.xpro.2021.101094 Code On Twitter
McCormick EM*, Arnemann KL*, Ito T, Hanson SJ, Cole MW (2022). "Latent functional connectivity underlying multiple brain states". Network Neuroscience; 6 (2): 570–590 doi:10.1162/netn_a_00234 [* = equal contribution] Code On Twitter
Schultz DH, Ito T, Cole MW (2022). "Global connectivity fingerprints predict the domain generality of multiple-demand regions". Cerebral Cortex, 32, 4464–4479 doi:10.1093/cercor/bhab495 On Twitter
Singh MF, Cole MW, Braver TS, Ching S (2022). "Developing control-theoretic objectives for large-scale brain dynamics and cognitive enhancement". Annual Reviews in Control. doi.org/10.1016/j.arcontrol.2022.05.001
Singh MF, Wang A, Cole MW, Ching S, Braver TS (2022). "Enhancing Task fMRI Preprocessing via Individualized Model-Based Filtering of Intrinsic Activity Dynamics". NeuroImage. doi:10.1016/j.neuroimage.2021.118836
2021
Hearne LJ, Mill RD, Keane BP, Repovs G, Anticevic A, Cole MW (2021). "Activity flow underlying abnormalities in brain activations and cognition in schizophrenia". Science Advances. 7(9) doi:10.1126/sciadv.abf2513 Code On Twitter
Mill RD, Winfield EC, Cole MW, Ray S (2021). "Structural MRI and functional connectivity features predict current clinical status and persistence behavior in prescription opioid users". NeuroImage: Clinical. 30. doi:10.1016/j.nicl.2021.102663
Keane BP, Barch DM, Mill R, Silverstein SM, Krekelberg B, Cole MW (2021). "Brain network mechanisms of visual shape completion". NeuroImage. 236. doi:10.1101/2020.08.03.233403
Cole MW, Ito T, Cocuzza C, Sanchez-Romero R (2021). "The functional relevance of task-state functional connectivity". Journal of Neuroscience. 41(12):2684-2702. doi:10.1523/JNEUROSCI.1713-20.2021 Code On Twitter
Sanchez-Romero R, Cole MW (2021). "Combining multiple functional connectivity methods to improve causal inferences". Journal of Cognitive Neuroscience. 33(2):180–194. doi:10.1162/jocn_a_01580 On Twitter Code
Spronk M, Keane BP, Ito T, Kulkarni K, Ji JL, Anticevic A, Cole MW (2021). "A whole-brain and cross-diagnostic perspective on functional brain network dysfunction". Cerebral Cortex. 31(1):547-561. doi:10.1093/cercor/bhaa242 On Twitter
2020
Singh MF, Braver TS, Cole MW, Ching S (2020). "Estimation and validation of individualized dynamic brain models with resting state fMRI". NeuroImage. 221. doi:10.1016/j.neuroimage.2020.117046
Ito T, Hearne LJ, Cole MW (2020). "A cortical hierarchy of localized and distributed processes revealed via dissociation of task activations, connectivity changes, and intrinsic timescales". NeuroImage. 221. doi:10.1016/j.neuroimage.2020.117141 On Twitter Code
Mill RD, Gordon BA, Balota DA, Cole MW (2020). "Predicting dysfunctional age-related task activations from resting-state network alterations". NeuroImage. 221. doi:10.1016/j.neuroimage.2020.117167 On Twitter Code
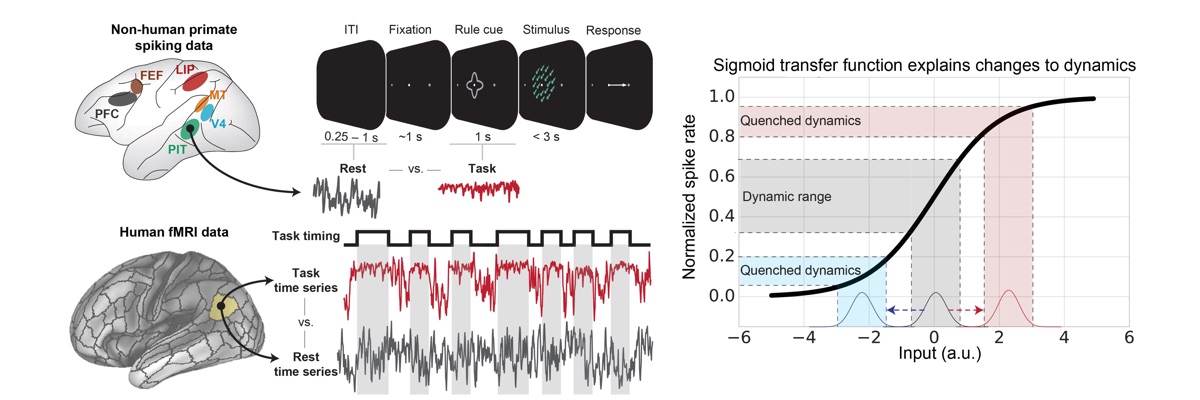
Ito T, Brincat SL, Siegel M, Mill RD, He BJ, Miller EK, Rotstein HG, and Cole MW (2020). "Task-Evoked Activity Quenches Neural Correlations and Variability in Large-Scale Brain Systems". PLOS Computational Biology. 16(8): e1007983. doi:10.1371/journal.pcbi.1007983 On Twitter
Cocuzza CV, Ito T, Schultz D, Bassett DS, Cole MW (2020). "Flexible coordinator and switcher hubs for adaptive task control". Journal of Neuroscience. 40(36):6949–6968. doi:10.1523/JNEUROSCI.2559-19.2020 On Twitter On Facebook Blog Post Code
Lamichhane B, Westbrook A, Cole MW, Braver T (2020). "Exploring brain-behavior relationships in the N-back task". NeuroImage. 212:1-11. doi:10.1016/j.neuroimage.2020.116683
Ito T, Hearne L, Mill R, Cocuzza C, Cole MW (2020). "Discovering the Computational Relevance of Brain Network Organization". Trends in Cognitive Sciences. 24, 25–38. doi:10.1016/j.tics.2019.10.005 On Twitter On Facebook
Kar K, Ito T, Cole MW, and Krekelberg B (2020). "Transcranial Alternating Current Stimulation Attenuates BOLD Adaptation and Increases Functional Connectivity". Journal of Neurophysiology. 123: 428–438. doi:10.1152/jn.00376.2019 On Twitter
2019
Reid A, Headley DB, Mill RD, Sanchez-Romero R, Uddin LQ, Marinazzo D, Lurie DJ, Valdés-Sosa PA, Hanson SJ, Biswal BB, Calhoun V, Poldrack RA, Cole MW (2019). "Advancing functional connectivity research from association to causation". Nature Neuroscience. doi: 10.1038/s41593-019-0510-4 On Twitter On Facebook
Yang GR, Cole MW, Rajan K (2019). "How to study the neural mechanisms of multiple tasks". Current Opinion in Behavioral Sciences. 29:134–143 doi.org/10.1016/j.cobeha.2019.07.001
Bolt T, Nomi JS, Bainter S, Cole MW, Uddin LQ (2019). “The Situation or the Person? Individual and Task-Evoked Differences in BOLD Activity”. Human Brain Mapping. doi:10.1002/hbm.24570
Cole MW, Ito T, Schultz D, Mill R, Chen R, Cocuzza C (2019). "Task activations produce spurious but systematic inflation of task functional connectivity estimates". NeuroImage. doi:10.1016/j.neuroimage.2018.12.054 On Twitter Code
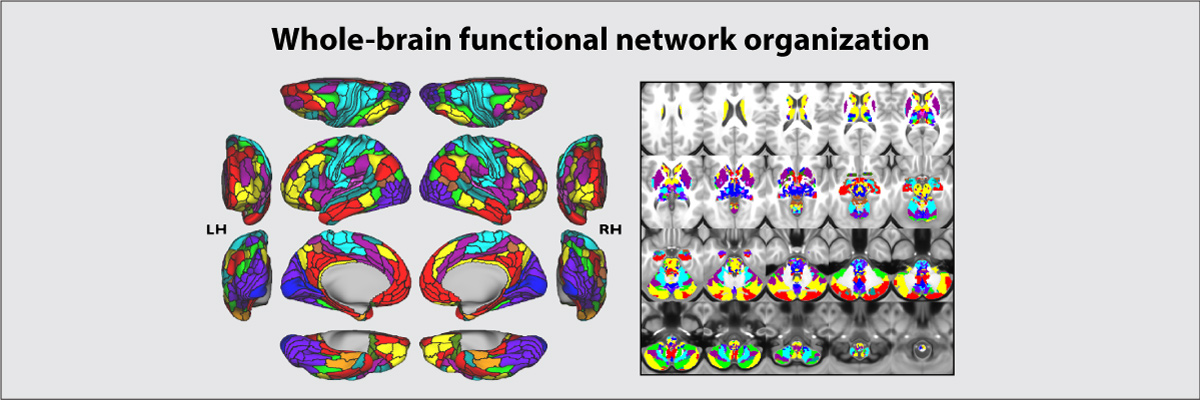
Ji JL*, Spronk M*, Kulkarni K, Repovs G, Anticevic A**, Cole MW** (2019). "Mapping the human brain's cortical-subcortical functional network organization". NeuroImage. 185:35–57. doi:10.1016/j.neuroimage.2018.10.006 [* = equal contribution; ** = senior authors] On Facebook Brain Network Partition
Schultz DH, Ito T, Solomyak L, Chen RH, Mill RD, Anticevic A, Cole MW (2019). "Global connectivity of the frontoparietal cognitive control network is related to depression symptoms in the general population". Network Neuroscience. 3:1:107-123. doi:10.1162/NETN_a_00056 On Twitter On Facebook Blog Post
2018
Chen RH, Ito T, Kulkarni KR, Cole MW (2018). "The human brain traverses a common activation-pattern state space across task and rest". Brain Connectivity. 8:7. doi:10.1089/brain.2018.0586 On Twitter On Facebook
Dixon ML, De La Vega A, Mills C, Andrews-Hanna J, Spreng RN, Cole MW, Christoff K (2018). "Heterogeneity Within the Frontoparietal Control Network and its Relationship to the Default and Dorsal Attention Networks". Proceedings of the National Academy of Sciences.
Cole MW, Patrick L, Meiran N, Braver T (2018). "A role for proactive control in rapid instructed task learning". Acta Psychologica. 184:20–30. doi:10.1016/j.actpsy.2017.06.004 On Twitter
2017
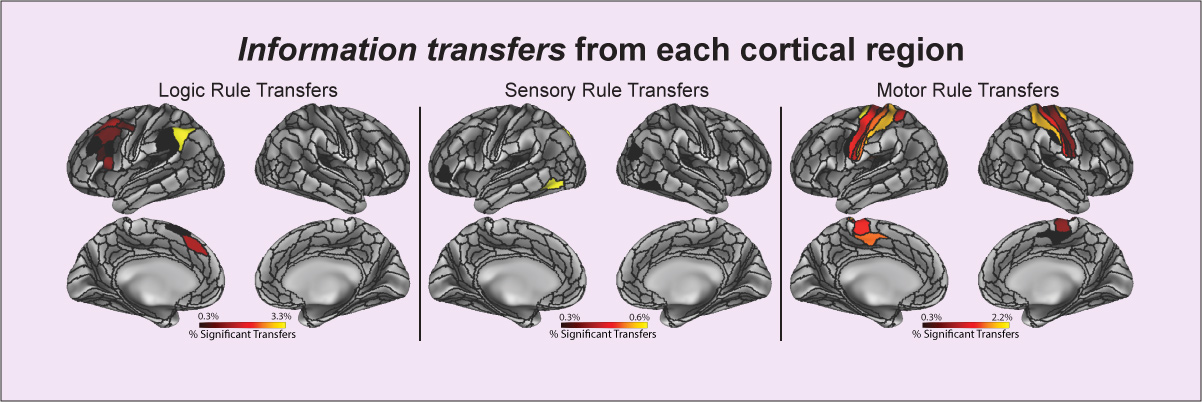
Ito T, Kulkarni KR, Schultz DH, Mill RD, Chen RH, Solomyak LI, Cole MW (2017). "Cognitive task information is transferred between brain regions via resting-state network topology". Nature Communications. 8:1027. doi:10.1038/s41467-017-01000-w On Facebook Blog Post Code
Cole, MW (2017). "Control and Connectivity: Dynamic Networks in the Human Brain". In The Wiley Handbook of Cognitive Control, edited by Tobias Egner, 314–33. Chichester, UK: John Wiley & Sons, Ltd. doi:10.1002/9781118920497.ch18.
Mill R, Ito T, Cole M (2017). "From connectome to cognition: The search for mechanism in human functional brain networks". NeuroImage. doi:10.1016/j.neuroimage.2017.01.060 On Twitter On Facebook
Cole MW, Braver T, Meiran N (2017). "The task novelty paradox: Flexible control of inflexible neural pathways during rapid instructed task learning". Neuroscience & Biobehavioral Reviews. doi:10.1016/j.neubiorev.2017.02.009 On Twitter On Facebook
Mill R, Bagic A, Bostan A, Schneider W, Cole MW (2017). "Empirical validation of directed functional connectivity". NeuroImage. 146:275–287. doi:10.1016/j.neuroimage.2016.11.037
2016
Cole MW, Ito T, Bassett DS, Schultz DH (2016). "Activity flow over resting-state networks shapes cognitive task activations". Nature Neuroscience. doi:10.1038/nn.4406 On Facebook Blog Post Code
Schultz DH & Cole MW (2016). "Higher Intelligence Is Associated with Less Task-Related Brain Network Reconfiguration". Journal of Neuroscience. doi:10.1523/JNEUROSCI.0358-16.2016
Schultz DH, Cole MW (2016) "Integrated Brain Network Architecture Supports Cognitive Task Performance". Neuron. 92:278–279. doi:10.1016/j.neuron.2016.10.004
Cole M.W., Yang G. J., Murray J. D., Repovs G., Anticevic A. (2016). "Functional connectivity change as shared signal dynamics". Journal of Neuroscience Methods. doi:10.1016/j.jneumeth.2015.11.011
New Method Simulations revealed that a version of correlation without variance normalization – covariance – was able to isolate differences in shared signal, increasing interpretability of observed functional connectivity change. Simulations also revealed cases problematic for non-normalized methods, leading to a “covariance conjunction” method combining the benefits of both normalized and non-normalized approaches.
Results We found that covariance and covariance conjunction methods can detect functional connectivity changes across a variety of tasks and rest in both clinical and non-clinical functional MRI datasets.
Comparison with Existing Method(s) We verified using a variety of tasks and rest in both clinical and non-clinical functional MRI datasets that it matters in practice whether correlation, covariance, or covariance conjunction methods are used.
Conclusions These results demonstrate the practical and theoretical utility of isolating changes in shared signal, improving the ability to interpret observed functional connectivity change.
Etzel J.A., Cole M.W., Zacks J.M., Kay K.N., Braver T.S. (2016). "Reward motivation enhances task coding in frontoparietal cortex". Cerebral Cortex. 26:4:1647-1659. doi.org/10.1093/cercor/bhu327
Cole M.W., Ito T., Braver T.S. (2016). "The behavioral relevance of task information in human prefrontal cortex". Cerebral Cortex. doi: 10.1093/cercor/bhv072
2015
Mattar M., Cole M.W., Thompson-Schill S., Bassett D.S. (2015). "A Functional Cartography of Cognitive Systems". PLOS Computational Biology. 11:12:e1004533. doi:10.1371/journal.pcbi.1004533.
Cole M.W., Ito T., Braver T.S. (2015). "Lateral prefrontal cortex contributes to fluid intelligence via multi-network connectivity". Brain Connectivity. doi: 10.1089/brain.2015.0357
Anticevic A., Hu X., Xiao Y., Hu J., Li F., Bi F., Cole M.W., Savic A., Yang G., Repovs G., Murray J., Wang X., Huang X., Lui S., Krystal J.H., and Gong Q. (2015). "Early-Course Unmedicated Schizophrenia Patients Exhibit Elevated Prefrontal Connectivity Associated with Longitudinal Change". Journal of Neuroscience. 35:1:267-286.
Meiran N., Pereg M., Kessler Y., Cole M.W., Braver T.S. (2015). "Reflexive Activation of Newly Instructed Stimulus-Response Rules: Evidence from Lateralized Readiness Potentials in NO-GO Trials". Cognitive, Affective, & Behavioral Neuroscience. 15:2:365-373.
Meiran N., Pereg M., Kessler Y., Cole M.W., Braver T.S. (2015). "The Power of Instructions: Proactive Configuration of Stimulus-Response Translation". Journal of Experimental Psychology: Learning, Memory, and Cognition. 41:3:768-786.
2014
Cole M.W., Bassett D.S., Power J.D., Braver T.S., Petersen S.E. (2014). "Intrinsic and task-evoked network architectures of the human brain". Neuron. 83(1), 238 – 251. doi:10.1016/j.neuron.2014.05.014
Yang G.J., Murray J.D., Repovs G., Cole M.W., Savic A., Glasser M.F., Pittenger C., Krystal J.H., Wang X., Pearlson G.D., Glahn D.C., Anticevic A. (2014). "Altered global brain signal in schizophrenia". Proceedings of the National Academy of Sciences. 111:7438–7443. doi:10.1073/pnas.1405289111
Cole M.W., Repovs G., Anticevic A. (2014). "The frontoparietal control system: A central role in mental health". The Neuroscientist, 20(6), 652–664. doi:10.1177/1073858414525995 On Twitter
Anticevic A., Hu S., Zhang S., Savic A., Billingslea E., Wasylink S., Repovs G., Cole M.W., Bednarski S., Krystal J.H., Bloch M.H., Li C.R., Pittenger C. (2014). "Global resting-state functional magnetic resonance imaging analysis identifies frontal cortex, striatal, and cerebellar dysconnectivity in obsessive-compulsive disorder." Biological Psychiatry, 75(8), 595–605. doi:10.1016/j.biopsych.2013.10.021
Obsessive-compulsive disorder (OCD) is associated with regional hyperactivity in cortico-striatal circuits. However, the large-scale patterns of abnormal neural connectivity remain uncharacterized. Resting-state functional connectivity studies have shown altered connectivity within the implicated circuitry, but they have used seed-driven approaches wherein a circuit of interest is defined a priori. This limits their ability to identify network abnormalities beyond the prevailing framework. This limitation is particularly problematic within the prefrontal cortex (PFC), which is large and heterogeneous and where a priori specification of seeds is therefore difficult. A hypothesis-neutral, data-driven approach to the analysis of connectivity is vital.
Methods
We analyzed resting-state functional connectivity data collected at 3T in 27 OCD patients and 66 matched control subjects with a recently developed data-driven global brain connectivity (GBC) method, both within the PFC and across the whole brain.
Results
We found clusters of decreased connectivity in the left lateral PFC in both whole-brain and PFC-restricted analyses. Increased GBC was found in the right putamen and left cerebellar cortex. Within regions of interest in the basal ganglia and thalamus, we identified increased GBC in dorsal striatum and anterior thalamus, which was reduced in patients on medication. The ventral striatum/nucleus accumbens exhibited decreased global connectivity but increased connectivity specifically with the ventral anterior cingulate cortex in subjects with OCD.
Conclusions
These findings identify previously uncharacterized PFC and basal ganglia dysconnectivity in OCD and reveal differentially altered GBC in dorsal and ventral striatum. Results highlight complex disturbances in PFC networks, which could contribute to disrupted cortical-striatal-cerebellar circuits in OCD.
Anticevic A., Tang Y., Cho Y. T., Repovs G., Cole M. W., Savic A., Wang F., Krystal J.H., and Xu K. (2014). "Amygdala Connectivity Differs Among Chronic, Early Course, and Individuals at Risk for Developing Schizophrenia". Schizophrenia Bulletin 40 (5): 1105-1116 doi:10.1093/schbul/sbt165.
Anticevic, A., Cole, M.W., Repovs, G., Murray J.D., Brumbaugh, M.S., Winkler, A.M., Savic, A., Krystal, J.H., Pearlson, G.D., & Glahn, D.C. (2014). "Characterizing Thalamo-Cortical Disturbances in Schizophrenia and Bipolar Illness". Cerebral Cortex. doi:10.1093/cercor/bht165
2013
Cole M.W., Reynolds J.R., Power J.D., Repovs G., Anticevic A., Braver T.S. (2013). "Multi-task connectivity reveals flexible hubs for adaptive task control". Nature Neuroscience; 2013 Sep;16(9):1348-55. doi:10.1038/nn.3470. Commentary on paper More info
Anticevic A., Cole M.W., Repovš G., Savic A., Driesen N.R., Yang G., Cho Y.T., Murray J.D., Glahn D.C., Wang X., and Krystal J.H. (2013). "Connectivity, Pharmacology and Computation: Towards a Mechanistic Understanding of Neural System Dysfunction in Schizophrenia". Front. Psychiatry 4:169. doi:10.3389/fpsyt.2013.00169
Anticevic A., Brumbaugh M.S., Winkler A.M., Lombardo L.E., Barrett J., Corlett P.R., Kober H., Gruber J., Repovs G., Cole M.W., Krystal J.H., Pearlson G.D., & Glahn D.C. (2013). "Global prefrontal and fronto-amygdala dysconnectivity in bipolar I disorder with psychosis history." Biological Psychiatry. 73(6): 565-573; doi:10.1016/j.biopsych.2012.07.031
Pathophysiological models of bipolar disorder postulate that mood dysregulation arises from fronto-limbic dysfunction, marked by reduced prefrontal cortex (PFC) inhibitory control. This might occur due to both disruptions within PFC networks and abnormal inhibition over subcortical structures involved in emotional processing. However, no study has examined global PFC dysconnectivity in bipolar disorder and tested whether regions with within-PFC dysconnectivity also exhibit fronto-limbic connectivity deficits. Furthermore, no study has investigated whether such connectivity disruptions differ for bipolar patients with psychosis history, who might exhibit a more severe clinical course.
Methods
We collected resting-state functional magnetic resonance imaging at 3 T in 68 remitted bipolar I patients (34 with psychosis history) and 51 demographically matched healthy participants. We employed a recently developed global brain connectivity method, restricted to PFC (rGBC). We also independently tested connectivity between anatomically defined amygdala and PFC.
Results
Bipolar patients exhibited reduced medial prefrontal cortex (mPFC) rGBC, increased amygdala–mPFC connectivity, and reduced connectivity between amygdala and dorsolateral PFC. All effects were driven by psychosis history. Moreover, the magnitude of observed effects was significantly associated with lifetime psychotic symptom severity.
Conclusions
This convergence between rGBC, seed-based amygdala findings, and symptom severity analyses highlights that mPFC, a core emotion regulation region, exhibits both within-PFC dysconnectivity and connectivity abnormalities with limbic structures in bipolar illness. Furthermore, lateral PFC dysconnectivity in patients with psychosis history converges with published work in schizophrenia, indicating possible shared risk factors. Observed dysconnectivity in remitted patients suggests a bipolar trait characteristic and might constitute a risk factor for phasic features of the disorder.
Cole M.W., Laurent P., Stocco A. (2013). "Rapid instructed task learning: A new window into the human brain’s unique capacity for flexible cognitive control". Cognitive, Affective, & Behavioral Neuroscience. 13(1): 1-22; doi:10.3758/s13415-012-0125-7
2012
Anticevic A., Cole M.W., Murray J., Corlett P.R., Wang X., & Krystal J.H. (2012). "The role of default network deactivation in cognition and disease". Trends in Cognitive Sciences. 16(12): 584–592; doi: 10.1016/j.tics.2012.10.008.
Cole M.W., Yarkoni T., Repovs G., Anticevic A., Braver T.S. (2012). "Global Connectivity of Prefrontal Cortex Predicts Cognitive Control and Intelligence". Journal of Neuroscience. 32(26): 8988-8999; doi: 10.1523/JNEUROSCI.0536-12.2012
Meiran N., Cole M.W., and Braver T.S. (2012). "When Planning Results in Loss of Control: Intention-Based Reflexivity and Working-Memory". Frontiers in Human Neuroscience. 6:104. doi: 10.3389/fnhum.2012.00104
2011
Cole M.W., Etzel J.A., Zacks J.M., Schneider W., Braver T.S. (2011). "Rapid transfer of abstract rules to novel contexts in human lateral prefrontal cortex". Frontiers in Human Neuroscience. 5:142. doi: 10.3389/fnhum.2011.00142
Cole M.W., Anticevic A., Repovs G., Barch D. (2011). "Variable global dysconnectivity and individual differences in schizophrenia". Biological Psychiatry 70(1):43-50. doi:10.1016/j.biopsych.2011.02.010
A fundamental challenge for understanding neuropsychiatric disease is identifying sources of individual differences in psychopathology, especially when there is substantial heterogeneity of symptom expression such as is found in schizophrenia. We hypothesized that such heterogeneity may arise in part from consistently widespread yet variably patterned alterations in the connectivity of focal brain regions.
Methods
We used resting state functional MRI to identify variable global dysconnectivity in 23 patients with DSM-IV schizophrenia relative to 22 age, gender, and parental socioeconomic status matched controls using a novel global brain connectivity (GBC) functional MRI method that is robust to high variability across individuals. We examined cognitive functioning using a modified Sternberg task and subtests from the Wechsler Adult Intelligence Scale - Third Edition. We measured symptom severity using the Scale for Assessment of Positive and Negative Symptoms.
Results
We identified a dorsolateral prefrontal cortex (DLPFC) region with global and highly variable dysconnectivity involving within-PFC under-connectivity and non-PFC over-connectivity in patients. Variability in this ‘under/over’ pattern of dysconnectivity strongly predicted the severity of cognitive deficits (matrix reasoning IQ, verbal IQ, and working memory performance) as well as individual differences in every cardinal symptom domain of schizophrenia (poverty, reality distortion, and disorganization).
Conclusion
These results suggest that global dysconnectivity underlies DLPFC involvement in the neuropathology of schizophrenia. Further, these results demonstrate the possibility that specific patterns of dysconnectivity with a given network hub region may explain individual differences in symptom presentation in schizophrenia. Critically, such findings may extend to other neuropathologies with diverse presentation.
2010
Cole M.W., Bagic A., Kass R., Schneider W. (2010). "Prefrontal Dynamics Underlying Rapid Instructed Task Learning Reverse With Practice". Journal of Neuroscience 30(42):14245-14254. doi: 10.1523/JNEUROSCI.1662-10.2010.
Cole M.W., Yeung N., Freiwald W., Botvinick M. (2010). "Conflict Over Cingulate Cortex: Between-Species Differences in Cingulate May Support Enhanced Cognitive Flexibility in Humans". Brain, Behavior, and Evolution 75(4): 239-240. doi: 10.1159/000313860. A response to Schall & Emeric, 2010.
Braver T.S., Cole M.W., Yarkoni T. (2010). "Vive les differences! Individual variation in neural mechanisms of executive control", Current Opinion in Neurobiology 20(2): 242-250. doi: 10.1016/j.conb.2010.03.002
Cole M.W., Pathak S., Schneider W. (2010). "Identifying the brain’s most globally connected regions", NeuroImage 49(4): 3132-3148. doi: 10.1016/j.neuroimage.2009.11.001
2009 and earlier
Cole M.W., Yeung N., Freiwald W., Botvinick M. (2009). "Cingulate cortex: Diverging data from humans and monkeys", Trends in Neurosciences 32(11): 566-574. doi: 10.1016/j.tins.2009.07.001
Cole M.W., Schneider W. (2007). “The cognitive control network: Integrated cortical regions with dissociable functions”, NeuroImage 37(1): 343-360. doi: 10.1016/j.neuroimage.2007.03.071
Schumacher E.H., Cole M.W., D’Esposito M. (2007). “Selection and Maintenance of Stimulus-Response Rules during Preparation and Performance of a Spatial Choice-Reaction Task”, Brain Research 1136(1): 77-87.
Hester R., D’Esposito M., Cole M.W., Garavan H. (2007). “Neural mechanisms for response selection: comparing selection of an item with a response from working memory”, NeuroImage 34(1): 446-54.
Curtis C.E., Cole M.W., Rao V., Ollinger J., D’Esposito M. (2005). “Canceling Planned Action: An fMRI Study of Countermanding Saccades”, Cerebral Cortex 15(9): 1281-9.
Conference Presentations
2025
Cole MW, Peterson KL, Chakravarthula L, Mill RD, Sanchez-Romero R (2025). Unveiling the cognitive relevance of functional connectivity through deconfounding. Poster at Cognitive Neuroscience Society,Boston, MA. Poster
Peterson KL, Hearne L, Mill RD, Cole MW (2025). The effects of aging on task representations during rapid instructed task learning. Poster at Cognitive Neuroscience Society, Boston, MA.
Tzalavras A, Osher D, Cocuzza C, Chakravarthula L, Mill RD, Peterson KL, Cole MW (2025). The human brain's functional connectome specifies hierarchical and non-hierarchical visual cortical processing. Poster at Cognitive Neuroscience Society, Boston, MA.
Aryal A, Mill RD, Amir I, Tzalavras A, Peterson KL, Salti M, Meiran N, Cole MW (2025). Cognitive control networks direct brain flows underlying rapid instructed learning of stimulus-response tasks. Poster at Cognitive Neuroscience Society, Boston, MA.
2023
Mill RD, Cole MW (November 2023). Neural representation dynamics reveal computational principles of cognitive task learning Poster at the Society for Neuroscience Annual Meeting, Washington, DC.
Peterson KL, Sanchez-Romero R, Mill RD, Cole MW (November 2023). Regularized partial correlation provides reliable functional connectivity estimates while correcting for widespread confounding Poster at the Society for Neuroscience Annual Meeting, Washington, DC.
Sanchez-Romero R, Chen R, Lalta N, Ito T, Mill RD, Cole MW (November 2023). Rapid learning to automaticity reveals learned content stored within patterns of resting-state functional connectivity changes Poster at the Society for Neuroscience Annual Meeting, Washington, DC.
Tzalavras A, Cocuzza C, Peterson KL, Chakravarthula LCN, Cole MW (November 2023). Brain network processes underlying the generation of hundreds of visual category responses in the human brain. Poster at the Society for Neuroscience Annual Meeting, Washington, DC.
2022
Mill RD, Flinker A, Cole MW (June 2022). Invasive human neural recording links resting-state connectivity to generation of task activity. Poster at the Organization for Human Brain Mapping (OHBM) conference, Glasgow, Scotland, UK.
Cocuzza CV, Mill RD, Cole MW (June 2022). The functional relevance of flexible hub connectivity in cognitive control networks. Poster at the Organization for Human Brain Mapping (OHBM) conference, Glasgow, Scotland, UK.
Singh MF, Wang C, Cole MW, Ching S (2022). "Efficient state and parameter estimation for high-dimensional nonlinear system identification with application to MEG brain network modeling". Proceedings from the American Control Conference. arXiv:2104.02827
Keane, B.P., Krekelberg, B., Mill, R.D., Silverstein, S.M., Thompson, J., Serody, M., Barch, D.M., & Cole, M. W. (May 2022). Compensatory brain network mechanisms of visual shape completion across the schizo-bipolar spectrum. Poster at Vision Sciences Society, St. Petersburg Beach, FL.
Keane, B.P., Krekelberg, B., Mill, R.D., Silverstein, S.M., Thompson, J., Serody, M., Barch, D.M., & Cole, M. W. (April 2022). Dorsal attention network dysfunction during visual perception in schizophrenia. Poster at Society for Biological Psychiatry, New Orleans, LA.
Keane, B.P., Krekelberg, B., Mill, R.D., Silverstein, S.M., Thompson, J., Serody, M., Barch, D.M., & Cole, M. W. (April 2022). Brain network mechanisms of visual perceptual organization in schizophrenia and bipolar disorder. Poster at Cognitive Neuroscience Society, San Francisco, CA.
2021
Cocuzza C.V., Mill R.D., Cole M.W. (November 2021). The functional relevance of flexible hub connectivity in cognitive control networks. Poster and group discussion H.04.c. on Network Activity at the Society for Neuroscience meeting, virtually.
Cocuzza CV, Raamana PR, Vilas M. (June 2021). 2021 open science special interest group discussion: Reproducible workflows.
Keane, B.P., Krekelberg, B., Mill, R.D., Silverstein, S.M., Thompson, J., Serody, M., Barch, D.M., & Cole, M. W. (November 2021). Brain network mechanisms of visual shape completion. Poster at Society for Neuroscience, virtually.
Ito T, Klinger T, Schultz DH, Cole MW, Rigotti M (February 2021). The role of compositional abstraction in human and artificial neural networks. Poster at Computational and Systems Neuroscience (Cosyne) conference, virtually.
Keane BP, Mill R, Krekelberg B, Barch D, Silverstein S, Cole MW (March 2021). Brain Network Mechanisms of Visual Shape Completion. Poster at the Cognitive Neuroscience Society (CNS) conference, virtually. Poster
2020
Hearne L, Mill R, Keane B, Cole MW (May 2020). Activity Flow Predictions Reveal the Role of Schizophrenia Network Abnormalities in Cognitive Activation and Behavioral Dysfunctions. Poster at the Biological Psychiatry conference.
Schultz DH, Ito T, Cole MW (June 2020). Cognitive control networks coordinate domain general task information throughout the brain. Poster #672 at the Organization for Human Brain Mapping (OHBM) conference, virtually.
Sanchez-Romero R & Cole MW (June 2020). Directed activity flow: Directed connectivity improves causal interpretation of predictive models. Poster #1753 at the Organization for Human Brain Mapping (OHBM) conference, virtually.
Hearne LJ, Mill RD, Keane BP, Cole MW (June 2020). Activity flow reveals the role of schizophrenia network dysfunction in cognition. Poster #87 at the Organization for Human Brain Mapping (OHBM) conference, virtually.
Mill RD, Hamilton J, Winfield EC, Lalta N, Chen RH, Spronk M, Cole MW (June 2020). Causal modeling of task information flow with high spatiotemporal precision in source EEG networks. Poster #1704 at the Organization for Human Brain Mapping (OHBM) conference, virtually.
Ito T, Hearne LJ, Cole MW (June 2020). Cognitive information differentiates between connectivity and activity across the cortical hierarchy. Poster #1397 and Oral Presentation at the Organization for Human Brain Mapping (OHBM) conference, virtually. Oral Presentation
2019
Schultz D.H., Ito T., Cole M.W. (October 2019). Cognitive control networks balance domain generality and specificity in representing task rule information across multiple cognitive domains. Poster at the Society for Neuroscience conference.
Mill R.D., Hamilton J.L., Winfield E.C., Lalta N., Chen R.H., Spronk M., Cole M.W. (June 2019). Decoding task information with high spatiotemporal prescision in source EEG networks. Poster presented at the Organization for Human Brain Mapping conference, Rome, Italy.
- fMRI insight into spatial signatures: interactions between sensory/motor content networks with higher-order cognitive control networks (CCNs i.e. DAN, FPN, CON; Ito et al., 2017).
- Primate multi-unit recordings (MUR) revealed complex dynamics (Siegel et al., 2015): task decodability is distributed across the brain, but with variability in decoding timecourse features (e.g. onset) elucidating regional specialization.
- However, limitations in fMRI temporal resolution and primate MUR spatial coverage have impeded characterization of both spatial and temporal task information signatures.
- We combined EEG source modeling and dynamic multivariate pattern analysis (MVPA) to decode task information with high spatiotemporal precision from large-scle human functional networks.
- We extended insight into "where" and "when" task information is represented with insight into "how"; using the activity flow process model (Cole et al., 2016) to predict future spatiotemporal dynamics from held-out regional activations transformed via resting-state connectivity lage motifs.
Ito T., Yang G.R., Cocuzza C.V., Schultz D.H., Cole M.W. (June 2019). Predicting motor behavior using neural encoding models during complex cognitive tasks. Poster presented at the Organization for Human Brain Mapping conference, Rome, Italy.
The brain integrates task contexts with sensory stimuli to produce motor behaviors during complex cognitive tasks. What is the neural implementation of stimulus-to-response transformations?
Hypothesis: Compositional neural representations of stimulus and task rule information can predict the outputs of a complex cognitive tasks
Cocuzza C.V., Ito T., Cole M.W. (June 2019). Intrinsic connectivity-based encoding models predict naturalistic visual representations. Poster presented at the Organization for Human Brain Mapping conference, Rome, Italy.
- Predictive encoding models benefit from condition-rich task paradigms
- Functional selectivity is thought to emerge from connectivity, but encoding models do not typically incorporate connectivity
- Task activity in held-out brain regions can be predicted by estimated activity flow over intrinsic connectivity patterns
- Question: What is the role of connectivity in the emergence of functionally specific neural activations?
- Hypothesis: Functionally specific neural activations representing cognitive features can be predicted by connectivity-based encoding models
Aim 1. Predict responses in fusiform face area (FFA) & parahippocampal place area (PPA) with connectivity-based encoding models
Purpose: establish connectivity model works for well-replicated FFA & PPA results
Data: HCP S1200[9] n-back working memory (WM) task; n=176
Purpose: extend connectivity model to condition-rich task paradigms
Data: HCP S1200[9] movie watching task (7T scanner); n=50
2018
Mill R.D., Gordon B.A., Balota D.A., & Cole M.W. (September, 2018). Predicting dysfunctional aging-related task activations from resting-state network alterations. Poster presented at the Resting State and Brain Connectivity conference, Montreal, Canada.
- Dementia has been linked to changes in fMRI task activations (Grady, 2012) and fMRI resting-state functional connectivity (rest FC; Ferreira & Busatto, 2013)
- Growing emphasis on identifying changes in these fMRI measures in ‘preclinical’ stages of dementia (Sheline & Raichle, 2013), given their potential use as functional imaging ‘biomarkers’ to speed up diagnosis and intervention
- Our understanding of these potential fMRI biomarkers has been hampered by segregated study of age-related task activation and rest FC alterations
- A strong relationship has been observed between these measures in young adults (Smith et al, 2009)
- Are age-related changes in fMRI task activation and rest FC measures underpinned by a common mechanism?
- Aim: extend ‘activity flow’ framework (Cole et al., 2016) to predict task activation dysfunction in preclinical aging
Cocuzza C.V., Hamilton J., Winfield E., Bassett D.S., Cole M.W. (September, 2018). A Network Science Cartography of Cognitive Control System Dynamics. Poster presented at Cognitive Computational Neuroscience, Philadelphia, PA.
- Estimate resting-state & task-evoked functional connectivity (rsFC, tFC), with high cognitive control task-sets that are highly dynamic5,7,15 from a large N human neuroimaging dataset
- Replicate prior findings that tFC closely resembles intrinsic rsFC6, with subtle, but functionally relevant, task-evoked reconfigurations1
- Assess reconfiguration properties of cognitive control networks (CCNs) using previously developed graph theoretical metrics
- Reconcile divergent properties and results with a novel metric, network partition deviation (NPD)
- Characterize CCNs using a cartographic approach that “maps” functional properties across multiple dimensions in 1 graph
- Use a split-sample validation approach to limit false discoveries
Ito T, Keane BP, Mill RD, Chen RH, Hearne LJ, Arnemann KL, He BJ, Rotstein HG, Cole MW (September, 2018). A dynamical systems model of intrinsic and evoked activity, variability, and functional connectivity. Poster presented at Conference on Cognitive Computational Neuroscience, Philadelphia, PA.
- Neural signals can be measured experimentally by estimating levels of acctivity, variability, and functional connectivity (FC), but these measurements are typically studied independently
- Previous studies have observed the relationship between activity and variability in theoretical models and empirical data (Abbot et al., 2011; He et al., 2011; Hennequin et al., 2018), but have not provided an explicit mechanism relating all three measurements
- Hennequin et al. demonstrated that an evoked stimulus drives a system to a stable fixed point, suggesting that neural dynamics can be studied with fixed point analysis from dynamical systems theory
We provide a dynamic network model to parsimoniously explain the emergence of activity, variability, and functional connectivity dynamics, while reproducing statistical phenomena widely described throughout the FC and neural activity literature
Ito T, Rotstein HG, Cole MW (July, 2018). A dynamical systems model of intrinsic and evoked activity, variability, and functional connectivity. Poster presented at Neurobiology of Cognition Gordon Research Conference, Newry, MA.
- Neural signals can be measured experimentally by estimating levels of acctivity, variability, and functional connectivity (FC), but these measurements are typically studied independently
- Previous studies have observed the relationship between activity and variability in theoretical models and empirical data (Abbot et al., 2011; He et al., 2011; Hennequin et al., 2018), but have not provided an explicit mechanism relating all three measurements
- Hennequin et al. demonstrated that an evoked stimulus drives a system to a stable fixed point, suggesting that neural dynamics can be studied with fixed point analysis from dynamical systems theory
We provide a dynamic network model to parsimoniously explain the emergence of activity, variability, and functional connectivity dynamics, while reproducing statistical phenomena widely described throughout the FC and neural activity literature
Ito T, Cole MW (June, 2018). Dimensionality of intrinsic network connectivity underlies flexible task representation. Poster presented at the Organization for Human Brain Mapping, Singapore.
- Intrinsic network organization forms the functional skeleton of the brain
- Task-evoked activations relate to neural processing related to task demands
- Strong correspondence between task activations and network organization (Smith et al. 2009)
- Task activations are shaped via intrinsic network organization (Cole et al., 2016; Ito et al., 2017)
Are there specific intrinsic network properties that ocntribute to the diversity of task-related patterns of activation associated with cognitive processing?
Mill, R.D., Gordon, B.A., Balota, D.A., & Cole, M.W. (June, 2018). Predicting dysfunctional aging-related task activations from resting-state network alterations. Poster presented at the Organization for Human Brain Mapping conference, Singapore.
- Dementia has been linked to changes in fMRI task activations (Grady, 2012) and fMRI resting-state functional connectivity (rest FC; Ferreira & Busatto, 2013)
- Growing emphasis on identifying changes in these fMRI measures in ‘preclinical’ stages of dementia (Sheline & Raichle, 2013), given their potential use as functional imaging ‘biomarkers’ to speed up diagnosis and intervention
- Our understanding of these potential fMRI biomarkers has been hampered by segregated study of age-related task activation and rest FC alterations
- A strong relationship has been observed between these measures in young adults (Smith et al, 2009)
- Are age-related changes in fMRI task activation and rest FC measures underpinned by a common mechanism?
- Aim: extend ‘activity flow’ framework (Cole et al., 2016) to predict task activation dysfunction in preclinical aging
2017
Ito T. & Cole M.W. (November, 2017). Cognitive control networks contain a mixture of diverse connectivity patterns characteristic of predicted flexible hub mechanisms. Poster presented at the Society for Neuroscience, Washington, DC.
Hypothesis: The diversity of a network's intrinsic connectivity drives its activation-based representational flexibility across a variety of tasks.
Schultz D., Ito T., Solomyak L., Chen R., Mill R., Kulkarni K., Cole M.W. (November, 2017). Systematic flexibility of global functional connectivity patterns supports flexible cognitive control. Poster presented at the Society for Neuroscience, Washington, DC.
Cole M.W. & Ito T. (September, 2017). Computational Network Mechanisms of Task-Evoked Functional Connectivity. Poster presented at Cognitive Computational Neuroscience, New York, NY.
Cole M.W., Schultz D., Mill R. (June, 2017). Activity flows over intrinsic and task-evoked functional networks shape cognitive task activations. Talk presented at the Organization for Human Brain Mapping, Vancouver, Canada.
Cole M.W., Ito T., Schultz D.H., Mill R.D. (March, 2017). Activity flows over task-evoked networks shape cognitive task activations across task switches. Poster presented at Cognitive Neuroscience Society, San Francisco, CA.
Ito T., Schultz D., Solomyak L., Chen R., Mill R., Cole M.W. (November, 2017). Cognitive control networks route task information to other networks via intrinsic functional connectivity pathways. Poster presented at the Society for Neuroscience, San Diego, CA.
Hypothesis: The diversity of a network's intrinsic connectivity drives its activation-based representational flexibility across a variety of tasks.
2016
Mill R., Bagic A., Schneider W., Cole M.W. (November, 2016). Network signatures of flexible cognitive control are reflected in oscillatory MEG source connectivity. Poster presented at the Society for Neuroscience, San Diego, CA.
- Rapid instructed task learnig (RITL) involves task reconfiguration of resting-state networks linked to cognitive control (e.g. frontoparietal network, FPN; Cole et al., 2010; Cole et al., 2013)
- fMRI's poor temporal resolution impedes insight into whether network control is a spectral i.e. frequency-specific phenomenon
- Animal electrophysiology suggests two beta band control processes
- Aim: Adapt MEG source functional connectivity (FC; Brookes et al., 2011; Hipp et al., 2012) to study on-task spectral features of resting-state networks
Schultz D., Ito T., Solomyak L., Chen R., Mill R., Kulkarni K., Cole M.W. (November, 2016). Cognitive control network global connectivity is related to the mental health of healthy individuals. Poster presented at the Society for Neuroscience, San Diego, CA.
Spronk M., Anticevic A., Cole M.W. (November, 2016). Cognitive control network flexible hub connectivity is altered across distinct mental illnesses. Poster presented at the Society for Neuroscience, San Diego, CA.
Ito T., Schultz D., Solomyak L., Chen R., Mill R., Cole M.W. (August, 2016). Intrinsic functional connectivity shapes task information between networks. Poster presented at the Neural Computation and Psychology Workshop, Philadelphia, PA.
Cole M.W., Schultz D., Chen R., Kulkarni K., Ito T. (April, 2016). The cognitive relevance of resting-state fMRI: Spontaneously organized networks and brain states across rest and task. Talk presented at Cognitive Neuroscience Society, New York, NY.
Ito T., Schultz D., Solomyak L., Chen R., Mill R., Cole M.W. (April, 2016). Flexible hub updates between tasks associated with global informational connectivity changes. Poster presented at Cognitive Neuroscience Society, New York, NY.
Chen R., Kulkarni K., Ito T., Cole M.W. (April, 2016). Spontaneously organized brain states revealed by dynamic multivariate pattern analysis of resting state fMRI. Poster presented at Cognitive Neuroscience Society, New York, NY.
- Distributed information-processing system with rich spatiotemporal dynamics
- Complex cognitive dynamics that map to the spatiotemporal dynamics
- Underlies neural basis of cognition
State space dynamics
- Visualize moment-to-moment whole brain activation patterns in a high dimensional feature space
- Apply MVPA and community detection algorithms to characterize this state space
We hypothesize that brain states (whole brain activation patterns) are organizaed in an hierarchical structure
- High level states - functionally general, reflective of basic cognitive functions
- Low level states - functionally specific depending on cognitive demands
- Mixture of task-positive and task-negative states in resting state, with more task-negative states being more dominant
Mill R., Bagic A., Schneider W., Cole M.W. (April, 2016). Empirical validation of directed functional connectivity across fMRI and MEG. Poster presented at Cognitive Neuroscience Society, New York, NY.
- The transition from 'undirected' (e.g. region A x B) to 'directed' (e.g. region A -> B) functional connectivity has been hampered by methodological issues... Algorithms? fMRI vs M/EEG? Parameters?
- Aim: To validate directionality methods in real fMRI and MEG data in the same subjects, via recovery of directed connectivity reversal in a 'sensory reactivation' paradigm
Schultz D., Cole M.W. (April, 2016). General intelligence and the efficiency of task-evoked brain network dynamics. Poster presented at Cognitive Neuroscience Society, New York, NY.
2015
Chen R, Shafto P, Cole MW (October 2015). Multivariate pattern analysis of resting state activity reveals spontaneously organized brain state dynamics. Poster presented at Society for Neuroscience, Chicago, IL.
- Neural activity at rest is nonstationary.
- Resting state network architecture present during task.
- Dynamic changes in functional connectivity using a popular sliding time window approach.
Multivariate pattern analysis (MVPA)
- Distributed activation patterns associated with cognition
- Commonly used in task neuroimaging
We hypothesize that spontaneous resting state dynamics are present in distributed multivariate activation pattern changes
Goals:
- Gain better understanding of resting state neural activity
- Expanding on the possible uses of MVPA
- Identify functionally meaningful brain states
Schultz DH, Cole MW (October 2015). Efficiency of brain network dynamics associated with cognitive ability. Poster presented at Society for Neuroscience, Chicago, IL.
Mill R, Bagic A, Schneider W, Cole MW (October 2015). Exploiting sensory reactivation from memory to validate directed functional connectivity measures with fMRI and MEG. Poster presented at Society for Neuroscience, Chicago, IL.
- The transition from 'undirected' (e.g. region A x B) to 'directed' (e.g. region A -> B) functional connectivity has been hampered by methodological issues... Algorithms? fMRI vs M/EEG? Parameters?
- Aim: To validate directionality methods in real fMRI and MEG data in the same subjects, via recovery of directed connectivity reversal in a 'sensory reactivation' paradigm
Cole MW, Bassett DS, Schultz DH (October 2015). Intrinsic and dynamic functional network architectures shape task-evoked activation patterns in the human brain. Poster presented at Society for Neuroscience, Chicago, IL.
Schultz DH, Cole MW (March 2015). Task-general and task-specifying functional brain dynamics. Poster presented at Cognitive Neuroscience Society, San Francisco, CA.
2014
Cole MW, Yang GJ, Murray JD, Repovs G, Anticevic A (November 2014). Reconceptualizing brain network change as shared signal dynamics. Talk presented at Society for Neuroscience, Washington DC.
Mattar MG, Cole MW, Thomspon-Schill SL, Bassett DS (November 2014). A dynamic functional cartography of cognitive systems. Poster presented at Society for Neuroscience, Washington DC.
2013
Cole MW, Bassett D, Power JD, Petersen S (November 2013). Mult-task functional connectivity reveals the human brain's dynamic network architecture and stable functional backbone. Poster presented at Society for Neuroscience, San Diego, CA.
2012
Cole MW, Reynolds JR, Power JD, Braver TS (October 2012). Flexible Hubs: A Novel Mechanism for Flexible Cognitive Control. Poster presented at Society for Neuroscience, New Orleans, LA.
Cole MW, Etzel J, Braver TS (April 2012). Identifying Flexible Hubs: A Novel Mechanism for Flexible Cognitive Control. Talk presented at Cognitive Neuroscience Society, Chicago, IL.
2011
Cole MW, Yarkoni T, Repovs G, Braver TS (November 2011). Flexible hubs: Global brain connectivity correlates of human intelligence. Talk presented at Society for Neuroscience, Washington DC.
Repovs G, Anticevic A, Cole MW, Barch DM (May 2011). Simulated comparisons of slow and rapid event-related task-based functional connectivity. Poster presented at Society for Biological Psychiatry; San Francisco, CA.
Cole MW, Yarkoni T, Repovs G, Braver TS (April 2011). Flexible hubs: Global brain connectivity correlates of human intelligence. Poster presented at Cognitive Neuroscience Society, San Francisco, CA.
2010
Cole MW, Braver T (November 2010). Task Set Formation: Switching to a Completely Novel Task Enhances Task Switching Costs. Talk presented at Psychonomics, St. Louis, MO.
Cole MW, Zacks J, Etzel JA, and Braver T (November 2010). Independent and distributed coding of task-set decision rules within prefrontal cortex. Poster presented at Society for Neuroscience, San Diego, CA.
Cole MW, Anticevic A, Repovs G, Barch D (August 2010). Locus of dysconnectivity: Dorsolateral prefrontol connectivity correlates with the cardinal symptoms of schizophrenia. Poster presented at the Gordan Research Conference: Neurobiology of Cognition, Waterville Valley, NH.
2009
Schneider W, Pathak S, Phillips J, Cole MW (2009). High Definition Fiber Tracking Exposes Circuit Diagram for Brain Showing Triarchic Representation, Domain General Control, and Metacognitive Subsystems. In Samsonovich, AV, Noelle, D, and Mueller, S (Eds.). Biologically Inspired Cognitive Architecture II: Papers from the AAAI Fall Symposium. AAAI Technical Report FS-09-01, Menlo Park, CA: AAAI Press.
Cole MW, Bagic A, Kass R, Schneider W (October 2009). Rapid Task Learning as a Window into the Neural Basis of Executive Control. Poster presented at Society for Neuroscience, Chicago, IL.
Cole MW, Schneider W (June 2009). From Symbols to Rules to Complex Behaviors: The Neural Basis of Rapid Instructed Task Learning. Poster presented at Human Brain Mapping, San Francisco, CA.
Cole MW, Pathak S, Schneider W (June 2009). Identifying the Brain's Most Globally Interactive Regions. Poster presented at Human Brain Mapping, San Francisco, CA.
2008
Cole MW, Kunkel A, Martins B, Schneider W (November 2008). The Neural Basis of Rapid Task Learning. Poster presented at Society for Neuroscience, Washington DC.
Pathak S*, Cole MW*, Schneider W (November 2008). Identifying the Brain's Most Globally Interactive Regions. Poster presented at Society for Neuroscience, Washington, DC. [*First two authors contributed equally]
Cole MW, Laurent P (November 2008). Neurevolution: An Example of Blogging To Enhance Scientific Communication. Poster presented at Society for Neuroscience, Washington, DC.
Cole MW, Martins B, Schneider W (April 2008). The Neural Basis of Rapid Instructed Task Learning. Poster presented at Cognitive Neuroscience Society, San Francisco, CA.
Pathak S, Martins B, Cole MW, Schneider W (April 2008). Anatomical and Functional Segmentation of the Cognitive Control Network: Supporting a preliminary cognitive control network connectome. Poster presented at Cognitive Neuroscience Society, San Francisco, CA.
Cole MW, Pathak S, Schneider W (April 2008). Medial Frontal Cortex Directs Attention Along Multiple Pathways to Resolve Perceptual Decision Difficulty. Poster presented at Cognitive Neuroscience Society, San Francisco, CA.
2007
Cole MW, Schneider W (June 2007). Perceptual Decision Making Is Mediated by the Cognitive Control Netowrk via ACC/pre-SMA to SLPFC Connectivity. Poster presented at Human Brain Mapping, Chicago, IL.
Cole MW, Schneider W (May 2007). Causal Connectivity Within a Cognitive Control Network During Perceptual Decision Making. Poster presented at Cognitive Neuroscience Society, New York, NY.
2006
Cole MW, Schneider W (June 2006). Dissociation of anterior cingulate, dorsolateral prefrontal, and fronto-polar cortex during a visual search task reveals specialized roles within a commonly activated fronto-parietal network. Poster presented at Human Brain Mapping, Florence, Italy.
Schneider W, Siegle G, McHugo M, Gemmer L, Jones D, Fissell K, Koerbel L, Suzuki I, Jung K, Goldberg R, Wheeler M, Cole MW, Hill N (June 2006). 2006 Pittsburgh Brain Activity Interpretation Competition: Inferring Experience Based Cognition from fMRI Data. Poster presented at Human Brain Mapping, Florence, Italy.
Cole MW, Schneider W (April 2006). Dissociation of anterior cingulate, dorsolateral prefrontal, and fronto-polar cortex during a visual search task reveals specialized roles within a commonly activated fronto-parietal network. Poster presented at Cognitive Neuroscience Society, San Francisco, CA.
2005
Cole MW, Schneider W (November 2005). Less Working Memory, More Control: Greater BOLD Response to Overcoming Prepotency in Prefrontal and Parietal Cortices. Talk presented at Society for Neuroscience, Washington DC.
Schneider W, Hill N, Cole MW (November 2005). Native and Supported Mode Processing in Attentional Control Network. Talk presented at Psychonomics, Toronto, Canada.
2004
Schneider W, Hill N, Chein J, McHugo M, Cole MW (November 2004). Subsystems Supporting attention, decision making, learning, and skilled performance. Talk presented at Psychonomics, Minneapolis, MN.
Schumacher EH, Cole MW, Singer A, D'Esposito M (October 2004). Distinguishing Response Selection Sub-processes with Functional Magnetic Resonance Imaging. Poster presented at Society for Neuroscience, San Diego, CA.
Schumacher EH, Cole MW, Singer A, D'Esposito M (April 2004). Distinguishing Response Selection Sub-processes with Functional Magnetic Resonance Imaging. Poster presented at Cognitive Neuroscience Society, San Francisco, CA.
Curtis CE, Cole MW, Rao V, Ollinger J, D'Esposito M (April 2004). Canceling planned action: An fMRI study of countermanding saccades. Poster presented at Cognitive Neuroscience Society, San Franscisco, CA.
2003
Curtis CE, Cole MW, Rao V, Ollinger J, D'Esposito M (October 2003). Canceling planned action: An fMRI study of countermanding saccades. Poster presented at Society for Neuroscience, New Orleans, LA.
Resources
The ColeNeuroLab_users email list
The Cole Neurocognition Lab software user group keeps users of Cole Lab GitHub public repositories (https://github.com/ColeLab) up to date with news about major updates & bugs. Users are also encouraged to post questions to this group, and to search through previous answers.
Analysis tools released by the Cole Neurocognition Lab
ActFlow Toolbox Toolbox meant to make it easier to implement the methods developed by Cole et al. (2016) and pursue the theoretical goals from Ito et al. (2020).
CombinedFC Code released as part of the project reported in Sanchez-Romero, et al. (Preprint)
Task FC Mean Activity Removal Code released as part of the project reported in Cole, et al. (2019)
The Cole-Anticevic Brain-wide Network Partition (CAB-NP) Brain atlas and code released as part of the project reported in Ji, Spronk, et al. (2019)
Information Transfer Mapping Code released as part of the project reported in Ito, et al. (2017)
ActFlowMapping Code released as part of the project reported in Cole, Ito, et al. (2016)
SimpleSims: A simple simulation framework for testing brain functional connectivity methods This code was developed as part of the project reported in Cole, Yang, et al. (2016)
Cole et al. (2013) Nature Neuroscience methods Code and FAQ for the Cole et al. (2013) paper
Datasets released by the Cole Neurocognition Lab
DynamicSMEGI This dataset contains whole brain task and resting-state high-density EEG data, along with a structural MRI for each participant (to aid in source localization). This dataset has been utilized in Mill et al. (2022).
Concrete Permuted Rule Operations This dataset contains whole brain task and resting-state multiband fMRI data for 96 subjects during a single recording session. Subjects performed the Concrete Permuted Rule Operations task (C-PRO). This dataset has been utilized in several studies, such as Ito et al. (2017), Cocuzza et al. (2020), Schultz et al. (2022), and Ito et al. (2022).
Participate in Our Research
Participants are being recruited for a variety of neuroimaging and behavioral studies conducted in our laboratory at the Center for Molecular and Behavioral Neuroscience (Rutgers University-Newark). Please email us for more info.
Address
Center for Molecular and Behavioral Neuroscience (Aidekman Research Center)Suite 212
197 University Avenue
Newark, NJ 07102